Difference between revisions of "Limonene"
| Line 153: | Line 153: | ||
[[Category:Biologically-derived compounds]] | [[Category:Biologically-derived compounds]] | ||
[[Category:Hydrocarbons]] | [[Category:Hydrocarbons]] | ||
| + | [[Category:Liquids]] | ||
Revision as of 22:39, 20 January 2020
 |
This article is a stub. Please help Sciencemadness Wiki by expanding it, adding pictures, and improving existing text.
|
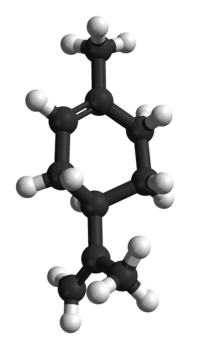
| |
| Names | |
|---|---|
| IUPAC name
1-Methyl-4-(1-methylethenyl)-cyclohexene
| |
| Other names
4-Isopropenyl-1-methylcyclohexene
Dipentene DL-limonene p-Menth-1,8-diene | |
| Properties | |
| C10H16 | |
| Molar mass | 136.24 g/mol |
| Appearance | Colorless or yellowish liquid |
| Odor | Orange-like |
| Density | 0.8411 g/cm3 |
| Melting point | −74.35 °C (−101.83 °F; 198.80 K) |
| Boiling point | 176 °C (349 °F; 449 K) |
| 0.0013 g/100 ml (20 °C) 0.00138 g/100 ml (25 °C) | |
| Solubility | Miscible in alcohol, benzene, carbon disulfide, carbon tetrachloride, chloroform, diethyl ether, oils Immiscible with propylene glycol |
| Vapor pressure | 1 mmHg (at 14 °C) 5 mmHg (at 40 °C) 10 mmHg (at 54 °C) |
| Thermochemistry | |
| Hazards | |
| Safety data sheet | Sigma-Aldrich (D-Limonene) |
| Flash point | 50 °C (122 °F; 323 K) |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Limonene is a colourless liquid hydrocarbon, classified as a cyclic terpene. The D-isomer is the most common form encountered. The name comes from the lemon, as many citrus fruits and some evergreen plants contain high amounts of the D-isomer. It may be recognized as the smell of oranges, lemons, or other citrus fruits.
Contents
Properties
Chemical
Limonene is a relatively stable terpene and can be distilled without decomposition, although at elevated temperatures it cracks to form isoprene.
Physical
Limonene is a colorless liquid hydrocarbon. It has a strong smell of orange or lemon peels.
Availability
Limonene can be extracted from citrus fruit peels via steam distillation.
Pure limonene can also be bought online.
Preparation
The synthesis of limonene from basic hydrocarbons is complex, instead it's more efficient to extract it from citrus fruit juice.[1] It is often prepared in introductory organic chemistry classes by steam distilling an orange peel.
Projects
- Extract various alkaloids from plants
Handling
Safety
Limonene and its oxidation products are skin and respiratory irritants, and limonene-1,2-oxide (formed by aerial oxidation) is a known skin sensitizer.
Storage
Should be stored under an inert gas, as it will slowly oxidize in contact with air.
Disposal
Limonene can be safely burned, however this produces lots of soot. Due to it's relative high flash point (50 °C) and to reduce soot, mix limonene with a more flammable solvent and then burn it. Best to do it outside.