Menthol
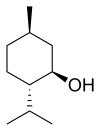
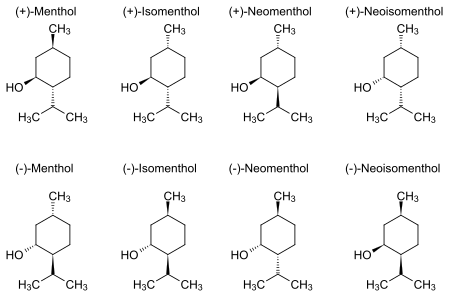
Menthol is an organic compound made synthetically or obtained from peppermint or other mint oils. It is a waxy, crystalline substance, clear or white in color, which is solid at room temperature and melts slightly above. The main form of menthol occurring in nature is ( − )-menthol, which is assigned the (1R,2S,5R) configuration. Menthol has local anesthetic and counterirritant qualities, and it is widely used to relieve minor throat irritation. Menthol also acts as a weak kappa Opioid receptor agonist.
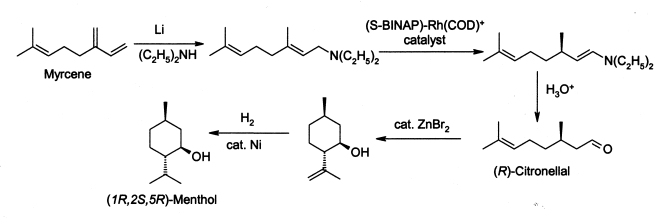
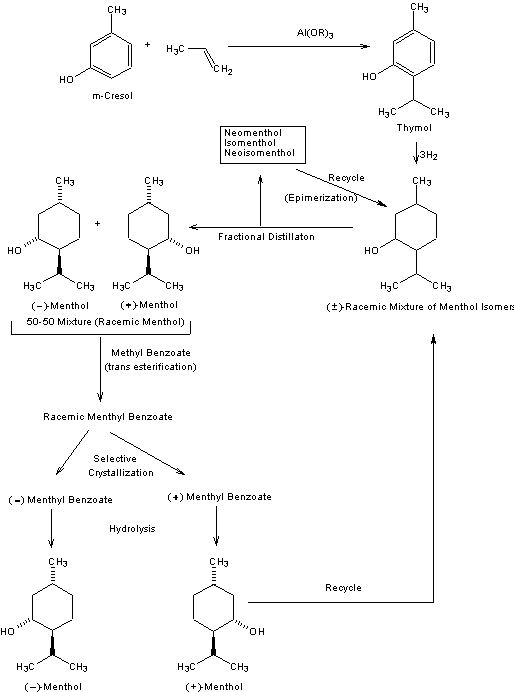
As with many widely-used natural products, the demand for menthol greatly exceeds the supply from natural sources. Menthol is manufactured as a single enantiomer (94% ee) on the scale of 3,000 tons per year by Takasago International Corporation. The process involves an asymmetric synthesis developed by a team led by Ryōji Noyori: