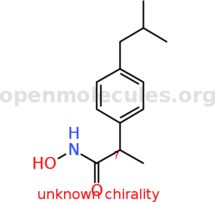
| It is important to use two moles of potassium hydroxide per mole of hydroxylamine hydrochloride. If only a 1:1 molar ratio is used, there is only a
small yield of hydroxamic acid formed as hydroxylamine itself is a relatively poor nucleophile. The reactive species in the reaction with esters is
not hydroxylamine but the much more nucleophilic hydroxylamine anion: |