Assured Fish
Hazard to Others
  
Posts: 319
Registered: 31-8-2015
Location: Noo Z Land
Member Is Offline
Mood: Misanthropic
|
|
Gamma-aminobutyric acid synthesis
So it has come to my attention as of late that GABA or Gama-aminobutyric acid is quite difficult to acquire in many parts of the world due to its more
nefarious uses.
This has had me thinking about ways in which we could easily synthesize it from more aquisitionable materials.
A few ideas sprung up with regards to decarboxylation of pyroglutamic acid followed by hydrolysis to open the ring, however the conditions for the
only paper i could find regarding this were rather extreme.
https://pubs.rsc.org/en/content/articlelanding/2015/gc/c4gc0...
Then today I had an epiphany, why not simply carry out a hofmann degradation of glutarimide.
We all know that we can do a similar reaction to prepare anthranilic acid from phthalamide so the mechanism should follow along the same pathway.
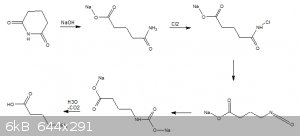
Now this is where it all falls short, in principle glutarimide could be prepared by thermolysis of ammonium glutarimate however ive not found a
readily available source for glutaric acid. Only 1,5-pentadiol which is from alfa.
https://www.alfa.com/en/catalog/A13998/
In principle any of these compounds should be fairly common and cheap yet little of it is available to the public.
So does anyone have any ideas as to where one could acquire either materials or a precursor to them?
Also can you think of any reason for this not working.
Sufficiently advanced science is indistinguishable from madness.
|
|
|
Pumukli
National Hazard
   
Posts: 686
Registered: 2-3-2014
Location: EU
Member Is Offline
Mood: No Mood
|
|
"Also can you think of any reason for this not working."
As I'm "enjoying" the unpredictableness of the synthesis of anthranilic acid / isatoic anhydride via the Hofmann route nowadays I read a bit about
the rearrangement too.
I remember that read in "Organic Reactions volume 3" that glutarimide is not following the expected route!
At least there was no report on a successfull Hofmann rearrangement with this compound up until 1947 when this book was sent to print. (page 274).
Maybe something happened during the past 72 years regarding this particular reaction but somehow I doubt it. 
You should first search for a successfull reaction with glutarimide! (A Reaxis search anyone?)
|
|
|
Corrosive Joeseph
National Hazard
   
Posts: 915
Registered: 17-5-2015
Location: The Other Place
Member Is Offline
Mood: Cyclic
|
|
Incoming........
/CJ
Attachment: REAXYS - 4-Aminobutanoic acid GABA (99 Reactions).pdf (1.2MB)
This file has been downloaded 483 times
Being well adjusted to a sick society is no measure of one's mental health
|
|
|
draculic acid69
International Hazard
    
Posts: 1371
Registered: 2-8-2018
Member Is Offline
|
|
I don't know about hard to get.and it's not well known for being "nefarious". everybody knows what pseudo ephedrine is mainly used for but ask anyone
who isn't into chemistry what gaba can be used to make and they don't have a clue because they've never heard of it.it is expensive though at $56 a
kilo.hard to get no expensive yes.
|
|
|