| Pages:
1
..
3
4
5
6 |
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
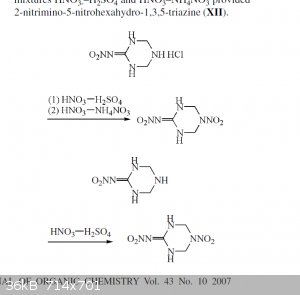
Looking at Metelkina again, I think the molecule is likely NNHT, 2-nitrimino-5-nitrohexahydro-1,3,5-triazine, m.p. 187 C. Metelkina found two routes
to this compound from NHHT one with AN/HNO3 and another with HNO3/H2SO4. It seems like it might be possible to get to it also by nitrate salt/H2SO4,
skipping the NHHT*HCl and just allowing a salt acid mix to stir for a much longer time.
That could be interesting because although I can find no performance data on NNHT, there is a claim that NHHT is 1.80 g/cc and 9000m/s vd, so
obviously NNHT will be more dense and higher performance than that. If it could be achieved with a NO3 salt/H2SO4 bath that would mean it's possible
to get >9000m/s on nitrate salt instead of HNO3 which would be pretty cool. NNHT is also better than NHHT in terms of having a 187 C mp which is
much preferable to 102 C.
|
|
|
underground
National Hazard
   
Posts: 692
Registered: 10-10-2013
Location: Europe
Member Is Offline
|
|
Are you sure that you got the above energetic material and not the fully nitrated? (N-H group being replaced with N-NO2 group)
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Quote: Originally posted by underground  | | Are you sure that you got the above energetic material and not the fully nitrated? (N-H group being replaced with N-NO2 group) |
I'm in no way sure. I think it is likely because mixed acid was used. The reaction was pretty intense though so I suppose it could be the fully
nitrated. The sample should be complety bone dry by now. I will try to mp
|
|
|
underground
National Hazard
   
Posts: 692
Registered: 10-10-2013
Location: Europe
Member Is Offline
|
|
In any way, i can see a really good future on EM with Hexamine and NQ. It needs for sure further investigation. NQ can easily be made cheaply in large
quantities with just urea, ammonium nitrate and H2SO4. I am pretty sure the H2SO4 for the QN dehydration can be boiled down to azeotropic and be
reused.
[Edited on 19-11-2022 by underground]
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Quote: Originally posted by underground  | In any way, i can see a really good future on EM with Hexamine and NQ. It needs for sure further investigation. NQ can easily be made cheaply in large
quantities with just urea, ammonium nitrate and H2SO4. I am pretty sure the H2SO4 for the QN dehydration can be boiled down to azeotropic and be
reused.
[Edited on 19-11-2022 by underground] |
That would be genius. I have heard you can boil H2SO4 in an Erlenmeyer flask with a small diameter opening and lightly stretch a piece of PTFE tape
over the opening and tape it, that PTFE supposedly acts as a membrane and can boil off water beyond the azeotrope. I have read this also works for
ethanol. -- I agree that NQ has a very yet-under-utilized, strong future. Maybe more than anything else. It has an entire range of materials with the
entire span of properties. Basic NQ is a very good explosive. I agree with Koch that NQ is misunderstood because of its low density morphology. But if
some better high bulk density techniques were found or if people used simple eutetic melts, density issues are easily overcome and the explosive is
high power.
Im trying to measure mp on the NQ+hex+HNO3. Burned some lightly twisted in some foil. It is very very fast.
Maybe there needs to be a NQ magic thread where all of these things get tested and the only commonality is it's all things you can do with NQ. It's a
wide energetic topic. It has so many interesting properties and compounds it forms with different behavior. NQ + Sulfamic acid is another one.
Attachment: Nitroguan IHE III Koch.pdf (4.1MB)
This file has been downloaded 183 times
Attachment: Nitroguan IHE IV Koch.pdf (2MB)
This file has been downloaded 157 times
Attachment: NITROGUAN HIGH BULK DENSITY.pdf (601kB)
This file has been downloaded 147 times
Attachment: Nitroguanidine AP sensitize.pdf (1.3MB)
This file has been downloaded 137 times
Attachment: Sulfamic guanidines.pdf (250kB)
This file has been downloaded 155 times
[Edited on 19-11-2022 by Hey Buddy]
|
|
|
underground
National Hazard
   
Posts: 692
Registered: 10-10-2013
Location: Europe
Member Is Offline
|
|
Consider biguanide too. Maybe a bit more exotic but it is still there.
Just now into my mind came the tetranitro glycoluril which made with urea and glyoxal but it is not used much because it hydrolyse with moisture due
to that urea structure, just like keto-RDX. I am pretty sure tetranitro glycoluril can be made too with guanidine in order to avoid that hydrolysis
problem that it comes from the urea structure.
Quote: Originally posted by Hey Buddy  |
That would be genius. I have heard you can boil H2SO4 in an Erlenmeyer flask with a small diameter opening and lightly stretch a piece of PTFE tape
over the opening and tape it, that PTFE supposedly acts as a membrane and can boil off water beyond the azeotrope. I have read this also works for
ethanol. -- I agree that NQ has a very yet-under-utilized, strong future. Maybe more than anything else. It has an entire range of materials with the
entire span of properties. Basic NQ is a very good explosive. |
Actually, thinking of it again, it may not even be necessary. You may dissolve the guanidine nitrate in H2SO4 then add your nitric acid into that mix,
then add your hexamine. You could possibly skip the guanidine nitrate to nitroguanidine reaction by using directly the nitroguanidine dissolved in
H2SO4.
Regarding of cooling the reaction, if we finally found any useful energetic from this route, a cooling system can be made using peltier modules. They
can go below -26.
[Edited on 19-11-2022 by underground]
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Quote: Originally posted by underground  |
Actually, thinking of it again, it may not even be necessary. You may dissolve the guanidine nitrate in H2SO4 then add your nitric acid into that mix,
then add your hexamine. You could possibly skip the guanidine nitrate to nitroguanidine reaction by using directly the nitroguanidine dissolved in
H2SO4.
[Edited on 19-11-2022 by underground] |
GuNO3 to NQ isn't a dehydration but a complex self-nitration to nitrimino, and so GuNO3 in H2SO4 can nitrate other materials in reaction. I dont know
how that would look but I have read articles about nitrating other materials with GuNO3.
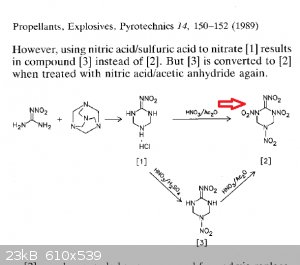
This material you asked to synth maybe the nitrimino version of Keto RDX. It is decomposing without melting at ~150 C. It vibrates on the hot plate
before decomp. The Nitrimino nitro Hexahydro mp is 187 C which is way above this material's decomp.
|
|
|
underground
National Hazard
   
Posts: 692
Registered: 10-10-2013
Location: Europe
Member Is Offline
|
|
Yea as i told you. It is logical because keto rdx with just urea is made with this way. I see no reason why it wont work with nitroguanidine. I am
sure that the material you just made may be even more energetic than HMX. Nitrimino keto-RDX has perfect OB, contains more nitrogen and it will be
more dense than the classic keto-RDX, Nitrimino keto-RDX will be even more powerful than classic keto-RDX. This is some serious stuff.
NNHT can possibly be made from the same reaction using diluted acids. When concentrated acids are used, nitrimino keto-RDX is produced. The material
needs further investigation.
The point is that even NNHT is quite energetic and it could possibly be made very easily with non concentrated acids or even with nitrate salt/ H2SO4
mix. Now if someone want to go further he can actually nitrate the NNHT to nitrimino keto-RDX. In any case it seems than NNHT is a really good
energetic by itself and an intermediate for nitrimino keto-RDX. On the other hand nitrimino keto-RDX seems to be the perfect EM. I really dont mind
if Nketo-RDX cannot be melt casted, you can always plasticize it or press it at high density for good performance.
Nketo-RDX seems to be the best energetic for using your precious Acids. Why would you waste them for RDX, classic keto-RDX ( hydrolysis problems) and
HMX (low yields and complicated synthesis)
Now if you want to go it even further, imagine the nitrimino diketo-HMX :-D
[Edited on 20-11-2022 by underground]
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Quote: Originally posted by underground  |
Nketo-RDX seems to be the best energetic for using your precious Acids. Why would you waste them for RDX, classic keto-RDX ( hydrolysis problems) and
HMX (low yields and complicated synthesis)
Now if you want to go it even further, imagine the nitrimino diketo-HMX :-D
[Edited on 20-11-2022 by underground] |
These molecules are all unexplored in literature and unfamiliar to readers. I would suggest that to avoid confusion, we should not refer to the
Nitrimino trinitrimine as "Nitrimino Keto RDX" because it's not a ketone at all. I dont know what the proper naming convention would be to suffice an
abbreviation, --It's still not for sure if mixed acid nitration from raw precursors yields this nitrimino. According to Zhuang, the zero oxygen
balance molecule decomps at 50 C. The material that I've prepared doesn't visibly decomp at 50 C, (I'm just using a PID hot plate, not a mp
apparatus), rather this prepared material starts jumping around~150 C.
Maybe a nickname for the zero OB compound is needed to distinguish it as a target from all the other subgroup molecules. Zero Oxygen Balance Nitrimino
RDX => Z RDX.
If Z RDX truly decomps at 50 C as Zhuang suggests. It probably cant be the material I've prepared because I dried it at ~55-60 C for 24 hours before
starting to play with it. It is very fast though. It does burn faster than any Hexamine derivative I've ever burned.
(lol, Zhuang writes "this makes [2] more powerful than RDX..." -yes it does.)

Also , we are diverging from Keto-RDX so this probably needs its own thread.
[Edited on 20-11-2022 by Hey Buddy]
"Now if you want to go it even further, imagine the nitrimino diketo-HMX :-D"
see image NQ+formaldehydenitration forms a di-nitrimino.

There is a possibility that the nitroso DNPT can be nitrimino'd, I have seen non-energetic tetrazocines that have two carbonyls and two nitros. So you
could possibly get di-nitrimino, di nitro/nitroso.
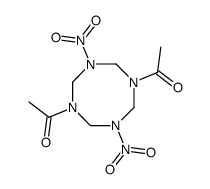
[Edited on 20-11-2022 by Hey Buddy]
|
|
|
underground
National Hazard
   
Posts: 692
Registered: 10-10-2013
Location: Europe
Member Is Offline
|
|
Interesting. Can you actually found any other file regarding ZRDX. Many times patents are wrong. Does it leave any residue when you burn it? If it is
Zrdx then there should not be any residue. I am still not 100% convinced that you have not made zrdx just because of the patent. keto-RDX is made with
the same way, i can not see any reason why not zrdx wont form. Maybe it could be a zrdx with just one NH group, something between NNTH and zrdx. I
would like to see also another patent describing zrdx and if they where identical then i could accept it.
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
I have only found a PEP article from 1989 on this compound they don't even bother naming it they just reference it by number. Without a standard
agreed name of the compound it's more challenging to cross reference it, you can only find it by stumbling upon it. All of this stuff I'm referencing
is from PEP -Zhuang &Yongzhong or for sub derivatives Metelkina who has done a lot of work on Nitroguanidines.
Honestly the only reason I'm in the ballpark of this compound is because of the NTNT molecule. The NTNT has a unique balance of CHNO and low-ish
sensitivity, casting and ease to manufacture where as something like say ZRDX is intensive and laborious to produce and is probably a bit more
sensitive.
Attachment: nitroguanidine poly nitro compounds Zhuang.pdf (283kB)
This file has been downloaded 126 times
Attachment: nitroguanidine derivatives.pdf (134kB)
This file has been downloaded 125 times
|
|
|
underground
National Hazard
   
Posts: 692
Registered: 10-10-2013
Location: Europe
Member Is Offline
|
|
Well, i see your point but the truth is that we have just scrach the surface on those substances. NNHT or even zrdx may be possible to synthesise with
nitrate salt and H2SO4. Most likely NNHT could be done with H2SO4 and azeotropic HNO3 wich is not that bad at all. On the other hand, sodium nitrite
is a bit expensive and i do not know any good procedure to make it while nitrate salt and H2SO4 is readily available.
I wish i could have the time and space to contribute to this whole project because i am also interested.
Anyway when you finally decided to do some performance tests compare to other traditional EM, i would like to give it a try to the above
BTW, Zrdx is probably called: 2-nitrimimo 1,3,5 trinitrotetrahydro 1,3,5 triazine
[Edited on 20-11-2022 by underground]
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
All good points. Nitrite does not have a good home production method, HNO3/NO3 does. Therefore HNO3/NO3 is actually more OTC than NO2. Guess an
acid/salt on the hexamine/NQ is in order. Has anyone tried bisulfate/nitrate in direct mix for nitration to bypass H2SO4? That would really be pushing
the OTC.
|
|
|
underground
National Hazard
   
Posts: 692
Registered: 10-10-2013
Location: Europe
Member Is Offline
|
|
Well, most likely reacting NQ and HEM with 65% nitric acid will make 2-nitrimino Triazine nitrate (Non nitrated). Now treating 2-nitrimino Triazine
nitrate with H2SO4 will make NNHT. Now further nitrating NNHT will make Zrdx.
Now it may be a bit difficult to isolate the nitrate salt since it will be most likely soluble in water.
By making Zrdx with the above method, rather than going directly for Zrdx, you are going to save quite a lot concentrated nitric acid and may be get
better yields since NNHT is already nitrated once. Now i dunno if the nitrate salt of NNHT is possible. If it is possible then nitrating the NNHT
nitrate to Zrdx would be even easier.
Also, most likely, you are going to avoid the run away problem since you do the nitration process step by step and not all at once.
Hey Buddy i am giving you food to experiment :p
NNHT itself would be quite energetic and from the above procedure it would be very easy to make without even the need of concentrated nitric acid. I
dunno if the synthesis can be made with nitrate salts since they may interact with the whole synthesis, especially ammonium nitrate. I would rather
choose to try sodium or potassium nitrate for the above synthesis.
[Edited on 21-11-2022 by underground]
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
I will certainly test what I can and try to video it. Here is a fun edit of the video log of your request experiment.
https://www.bitchute.com/video/W4qzvX3J7Bbj/
|
|
|
underground
National Hazard
   
Posts: 692
Registered: 10-10-2013
Location: Europe
Member Is Offline
|
|
Wow, nice video Buddy, i am impressed. Thanks, i appreciate it.
Edit. Maybe there is some traditional keto rdx there and when you dump it into the water it hydrolysed. I am not 100% sure though. Maybe also when you
dump it all at once some spots of really high temperature where created causing the product or nitric acid to decompose. If you try the synthesis
again, try to stir the water ice bath and poor it slowly. If this solve the problem then most likely was some high temp spots causing the product or
the nitric acid to decompose.
It seems from that file that Zrdx (NTNHT) goes back to keto-RDX with the presence of water, i am not 100% sure though since i dont know Chinese. So
maybe you got ZRDX (NTNHT) but it was hydrolysed upon contact with water to keto-RDX. Unfortunately the pdf is on Chinese. Nitrourea is unstable with
water as opposed to nitroguanidine so most likely yields would be very high compared to nitrourea. The subject needs further investigation for sure.
http://sioc-journal.cn/Jwk_yjhx/EN/Y2012/V32/I01/75
Even if NTNHT is unstable, NNHT is still promising since it should be made with diluted NA and SA. The Chinese pdf file has really interesting
synthesis and information about all those energetics. If anyone who knows Chinese could translate it, it would be really helpful. The perchloric salt
of NNHT may be exist since perchloric acid is a really strong acid. The perchloric salt of NNHT would still got zero OB and most likely be superior to
HMX. Even the perchloric salt of NIHT would be really powerful too. NIHT perchlorate dont even need nitric acid to produce.
[Edited on 22-11-2022 by underground]
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
underground. I didnt realize NIHT is the same compound as NHHT. Both are referred in research but they are the same. Once I realized it can be called
NIHT i started looking and found an old file describing an NIHT.HCL and it has instruction to prepare it in a closed vented vessel so that oxygen
doesnt contaminate reaction. Room temp reaction. The yield seems pretty high. I tried NIHT in an open vessel under other instruction on small-scale
with no yield so I will try this other method.
|
|
|
underground
National Hazard
   
Posts: 692
Registered: 10-10-2013
Location: Europe
Member Is Offline
|
|
Nice. Looking forward to your reply. Also could you try the Zrdx synthesis when you got time but use aminoguanidine instead of nitroguanidine?
[Edited on 6-12-2022 by underground]
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Sure, im trying to reduce NQ right now but I didnt know power supply was fried before I got the NQ in solution. Hopefully will recover AGu from
catholyte when new power supply shows up. Either way, I can do it when I get some AGu built up. Going to work on NIHT since it defeated me before, and
maybe a NH4NO3/H2SO4 bath on NQ+Hexamine.
|
|
|
MineMan
National Hazard
   
Posts: 996
Registered: 29-3-2015
Member Is Offline
Mood: No Mood
|
|
Updates from hey buddy using the aminoguanidine?
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Sorry. I took on waaaay too many projects like the newb I am. Practically exploring the king primaries right now. It's not going very smoothly. I am
trying to reproduce the NiBisAgu(ClO4)2 but currently trying to figure out better routes to AGu. Right now testing AGu from GuNO3+N2H4+NH4NO3. Still
not entirely clear though. I dont know a good way of verifying if AGu is produced other than proceeding on to reactions with it. Slow. I did find a
very cool way of producing small batch lead azide. But it needs a lot of work up to discuss, because it uses small scale HN3 and I just dont want to
recommend that to an unsuspecting reader. But it's a lot simpler IMO than isopropyl nitrite method. Anyways, I backed off the old primaries back to
AGu but everything got really slowed down with cold weather. Havent felt motivated. I will work on this soon though. It's not terribly laborious. The
sample made from Hex+NQ is very reactive to flame decomposition. It seems very powerful to me. I need to test it on a hammer strike. But havent go
around to it yet. Hopefully soon...
|
|
|
MineMan
National Hazard
   
Posts: 996
Registered: 29-3-2015
Member Is Offline
Mood: No Mood
|
|
Why NiBi? Never heard of that. Only the nickel one. AGu is easily bought online and ships fast.
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Sorry, i was just writing "Bis". Not bismuth. I didnt know you could buy AGu. I was trying to make it from GuNO3 because in US you can order that
nitrate for ~$10/lb
|
|
|
MineMan
National Hazard
   
Posts: 996
Registered: 29-3-2015
Member Is Offline
Mood: No Mood
|
|
You can buy the HCL as a supplement. Or the bicarbonate off of eBay. Use to be easier to buy. Strange how things only get less available on the web.
It is an impressive primary. The power is insane.
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Quote: Originally posted by MineMan  | You can buy the HCL as a supplement. Or the bicarbonate off of eBay. Use to be easier to buy. Strange how things only get less available on the web.
It is an impressive primary. The power is insane. |
I looked today but it seems relatively expensive for now. It is difficult to produce. IMO electrolytic reduction on lead is the most efficient method,
acetic acid/zinc acetate reductions are just a mess. Im confident hydrazine hydrate with guanidine produces AGu, I just cant find it in literature, so
Im doing experiments now trying to get the purple color of copper Agu ClO4, simply because it seems a little easier to ID if it gets formed versus the
nickel. If I cant find a simple production of AGu, there is also the metal primary salts of ANQ both nitrates and perchlorates. ANQ is low yield by
NQ+hydrazine, but it is reliable. There is also nitriminotetrazole salts from ANQ, those are practically unexplored. Surely there are some powerful
primaries there. Honestly, in everything ive tried, I begin to appreciate lead(ii) azide. It is toxic but there is really no comparison in character
and the body of historical literature behind it. For primary explosives that are realistic and reproduceable at scale on demand, it has a lot going
for it.
--Im also somewhat mesmerized by organic peroxides. Im under no delusion they can be likely tamed, but it seems probable. Most of the work with them
is only a few decades old, mostly minimal effort amateur stuff, indicative of quick organic primaries. But just to parse the matter at a strategic
level, in terms of primary explosives, it's very narrow sport. Minimal preparation complication is an exponential advantage before even the character
and performance. The threshold of success of a primary explosive is to simply initiate, that technical feat is low-threshold compared to necessities
of high performance secondaries. Something like a peroxide if it could be tamed down and made more usable would be advantageous due to preparation
simplicity. --Anyways, there are very good reasons that lead azide is king and is still unthreatened by new primaries. It simply works technically,
like a raped-ape, and has industrial procedure polished.
[Edited on 5-1-2023 by Hey Buddy]
[Edited on 5-1-2023 by Hey Buddy]
|
|
|
| Pages:
1
..
3
4
5
6 |