12AX7
Post Harlot
    
Posts: 4803
Registered: 8-3-2005
Location: oscillating
Member Is Offline
Mood: informative
|
|
Simpler Gregar extractor
While staring at something I have warmed on the stove, I thought of the condensation, the distillation, then the extraction.. hmm Gregar extractor?
How could you rearrange that thing... aha! Simple enough that surely it's been posted here before, but I'm wondering if it has?
There's been enough chat about said extractor that I think I'll post this.
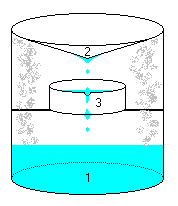
Take a closed jar, or tub, or whatever, with solvent 1 in it. Add heat (warm stove, sun, hotplate..), solvent evaporates and condenses on 2, which
has a cooled cone on top (like the "survival still" plan). Condensate drips into container 3 (optionally set on a stand), holding sample to
be extracted. As distillate fills 3, it overflows into 1.
I figure this would be a good way to do things if you have a lot of time to wait for something... sit it in the sun and forget about it for a few
weeks. It would of course go faster on a hotplate and/or with cooling air or water on the top.
Depending on whether or not your materials are corrosive or require high heat, you could use wood, plastic, metal, glass, whatever you want.
Tim
|
|
|
Fleaker
International Hazard
    
Posts: 1252
Registered: 19-6-2005
Member Is Offline
Mood: nucleophilic
|
|
Hm, I would make part 3 a narrow-mouthed vessel to less evaporation and also immerse that in the substance being evaporated so it does not condense on
the bottom of part 3.
|
|
|
FrankRizzo
Hazard to Others
  
Posts: 202
Registered: 9-2-2004
Member Is Offline
Mood: No Mood
|
|
That's more soxhlet-based than Gregar (improved soxhlet), but a nice hack if you've got minimal resources and a bunch of time.
|
|
|
12AX7
Post Harlot
    
Posts: 4803
Registered: 8-3-2005
Location: oscillating
Member Is Offline
Mood: informative
|
|
Ah, so it is...(I don't know why I haven't looked up Soxhlet until now). They are pretty similar, it just looks like Gregar runs
continuously or dripwise rather than toilet-flush-wise (  , well it does say
siphon on the Soxhlet, so it must have a lot more hysteresis). , well it does say
siphon on the Soxhlet, so it must have a lot more hysteresis).
Yeah, a narrow mouth would be better, and the whole thing would work much better in a squat container, if you can get one.
Tim
|
|
|
Quince
National Hazard
   
Posts: 773
Registered: 31-1-2005
Location: Vancouver, BC
Member Is Offline
Mood: No Mood
|
|
The other difference, besides being continuous, is that the Gregar can be reconfigured to do liquid/liquid extractions as well, with either heavier or
lighter solvent.
They show the three configurations here:
http://chemistry.anl.gov/preview/GregarWeb/GE-main.html
\"One of the surest signs of Conrad\'s genius is that women dislike his books.\" --George Orwell
|
|
|