| Pages:
1
2
3 |
smaerd
International Hazard
    
Posts: 1262
Registered: 23-1-2010
Member Is Offline
Mood: hmm...
|
|
Simplest Colorimeter
So I wanted a little colorimeter just for kicks and maybe to run a couple basic experiments. I was eyeing out op-amps and photodiodes, then I hit the
literature. I ran into this wonderful little article -
"A Simple, Small-Scale Lego Colorimeter with a Light-Emitting Diode (LED) Used as Detector. Jonas Asheim, Eivind V. Kvittingen, Lise Kvittingen, and
Richard Verley
Journal of Chemical Education 2014 91 (7), 1037-1039. DOI: 10.1021/ed400838n "
This is probably the simplest design for a colorimeter I've ever seen, although I do not know it's linear ranges, it seems to work okay for basic
things. Basically they have a LED light source driven by a battery or in my case a wall-charger, which shines through a cuvette, and is detected by a
Red LED.

The authors found like I had in a previous experiment (http://www.sciencemadness.org/talk/viewthread.php?tid=53814#...) that LED's can serve as detectors for photons of wavelengths of light up to what
they emit. Ex: a 570nm LED can detect photons from 0nm->570nm (okay maybe not 0nm but you know what I mean).
Anyways, it was such a simple experiment I had to try it myself and I was skeptical that the linear range of voltage incurred by illumination would
not be reproducible. Needless to say I was surprised.
Experiment - A 505nm LED was used as the light source and a 590nm LED was used as the detector. A 5v 500mA power supply was put in series with a
270ohm resistor and the source LED. 1 drop of green food coloring was solvated in ~10mL of water. then measured. The solution was diluted in half 4
times relative to the unknown initial concentration. Each time the solutions voltage was measured.
That's basically it, then I created %transmittance values and converted to absorbance and made a beer lambert law plot. The linear correlation
coefficient was 0.998, indicative of an acceptable fit.
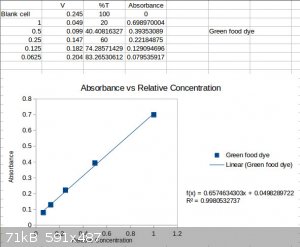
Try it for yourselves, its pretty nifty. I figured this might come in handy for some people needing to do some basic analysis or explain the principle
of visible light spectroscopy to someone.
Attachment: colorimeter results.ods (47kB)
This file has been downloaded 791 times
With this simple design I could envision a 10$-20$ variant, which includes a single amplification stage to suite a milivolt/volt display(if a suitable
display/IC could be found). So it would be without a microcontroller and without a multimeter. The math would still be required but it's really not a
big deal with an Excel file.
So it's not a UV-VIS but for someone who is broke and wants to experiment with kinetics of visible light absorbing compounds, LED's are dirt cheap.
Molar absorptivities could likely be calculated to about 2 decimals if the solution concentration was in an acceptable range(demonstrated by the
article).
[Edited on 28-5-2015 by smaerd]
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Yep, that seems to work really, well. Well done!
Igor (aga), please build me one of those, presto!
|
|
|
smaerd
International Hazard
    
Posts: 1262
Registered: 23-1-2010
Member Is Offline
Mood: hmm...
|
|
It's a 10 minute build blogfast. All you need are two lego blocks (the kind with the holes in them), two LED's, a resistor, batteries, and a
multimeter. Oh and something transparent to hold the solution in.
It would be cool if Aga built an amplification stage for this though. If not I might, but I am about to be running out of free-time here pretty soon I
think :/.
[Edited on 28-5-2015 by smaerd]
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
Arduino, three leds, CdS cell as sensor.
PWM the three LEDS to generate a full spectrum, also measurings the resultings.
Edit :
What's it for ?
To see the Colour of stuff ?
[Edited on 28-5-2015 by aga]
|
|
|
smaerd
International Hazard
    
Posts: 1262
Registered: 23-1-2010
Member Is Offline
Mood: hmm...
|
|
Aga, maybe with an RGB LED, other-wise the path-length of the light would be pretty skewed. Unless you did 3 detectors. I'm more of a fan of the LED
light source swapping so you can buy a LED catered to a wavelength of interest.
Do those CdS or PbS sensors have a linear response between voltage/resistance and illumination? And I guess are they sensitive in the visible range? a
phototransistor or diode would work as well.
It's for quantitation of a dissolved material at a known wavelength, or studying how much of a certain wavelength of light is absorbed per unit of
compound.
It would be easy to do on a microcontroller but I feel like that might hold someone back from building one, and add cost when a purely analog circuit
would suffice. Then again on a microcontroller an amplification stage becomes a likely necessity with the LED detector at least.
[Edited on 28-5-2015 by smaerd]
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
Needs an Arduino, TFT shield and a PIC as far as i just sketched out on this bit of paper.
Cost should be about $30 max.
Yes, i was thinking of an RGB LED as i have some.
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Nope. It's a simple quantitative analytical instrument which allows to determine the concentration of coloured reagents (or reagents that can be
'given' colour) by their absorbance in solution. Principle:
http://en.wikipedia.org/wiki/Beer%E2%80%93Lambert_law
10s and 10s and 10s of published analytical protocols based on this method, BTW.
I can see a bit of improvement to smaerd's set up by means of a shielded cuvette holder (no stray light).
[Edited on 28-5-2015 by blogfast25]
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
Sample in a test tube, dropped into a bit of copper pipe through which a hole has been drilled (laterally, all the way through = an in-hole and an
out-hole)
Ok. Think we have that capability.
Edit
It's the Beer-Lambert Law.
Unfortunately i have no Lambert, however i do have ....
[Edited on 28-5-2015 by aga]
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by aga  | Sample in a test tube, dropped into a bit of copper pipe through which a hole has been drilled (laterally, all the way through = an in-hole and an
out-hole)
|
NO TEST TUBE. Cuvette has to be square, incident light perpendicular to side. Test tube would reflect light in the wrong directions, causing false
readings. Trust me, I'm a Leprechaun.
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
So, seeing as i have copper pipe and no small plastic pipe, here's the Plan :-
Cut some 15mm copper pipe to a length a bit longer than one of my test tubes.
Drill a hole all the way through about half way down.
Drill a copper-pipe sized hole in another, shorter length of copper pipe.
Cut that one in half.
Solder the two halves onto the longer tube so that they cover the holes in the long bit, forming a kind of cross.
Stick an RGB LED onto one of the short tubes, a detector into the other.
Vary the RGB intensity to generate a rainbow spectrum.
Measure what intensity the sensor sees.
Do a graph and shout Hoorah !
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
Duh.
Damned leprechauns throwing spanners in the works.
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Square cuvettes are cheap, BTW.
I'm Leprechaun Lambert.
[Edited on 28-5-2015 by blogfast25]
|
|
|
smaerd
International Hazard
    
Posts: 1262
Registered: 23-1-2010
Member Is Offline
Mood: hmm...
|
|
Quote: Originally posted by blogfast25  |
NO TEST TUBE. Cuvette has to be square, incident light perpendicular to side. Test tube would reflect light in the wrong directions, causing false
readings. Trust me, I'm a Leprechaun. |
That may sound like a concern in theory, but many UV-VIS instruments utilize test-tubes or cylindrically shaped sample cells in their instruments. The
readings are relative to a blank. As long as the test-tube is placed in the same location any internal reflectance or similar phenomena will be
accounted for.
@aga - I guess I'm just confused what you're trying to accomplish by doing that. Are you going to have your light source in direct contact with the
solution? That might really limit things. Edit - Why are you varrying the intensity of the light?
[Edited on 28-5-2015 by smaerd]
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
All smaerd's design needs is to enclose optical part in a little light-tight box.
[Edited on 28-5-2015 by blogfast25]
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
Not got any.
So, Flat is just the surface of a sphere with an infinite radius, so perhaps a bit of curvyness is ok.
In any case, the LED light source, electronics, detector and software will be the same.
May as well do those bits and try out the test tube.
If that doesn't work i'll chop a lump off some polycarbonate sheeting that has square cells.
If that fails, best ebay some square glass.
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Igor, you will do as told, FOR ONCE, remember the Frankenstein II debacle, I DON'T want to go through that again! 
Cuvettes from FleeceBay.
[Edited on 28-5-2015 by blogfast25]
|
|
|
smaerd
International Hazard
    
Posts: 1262
Registered: 23-1-2010
Member Is Offline
Mood: hmm...
|
|
Blogfast - the little thing I made doesn't seem to be effected by stray light. The stray light is probably outside of the detection of the LED and I
got a pretty reasonable R^2 given the 'significant digits' I used. If the signal was amplified though, then that would be much more of a concern.
Edit - It's not like I broke out the volumetric glass-ware either. My solution measurements were made entirely via a disposable plastic pipett with
half mL graduations.
[Edited on 28-5-2015 by smaerd]
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
A picture speaks a 1000 words
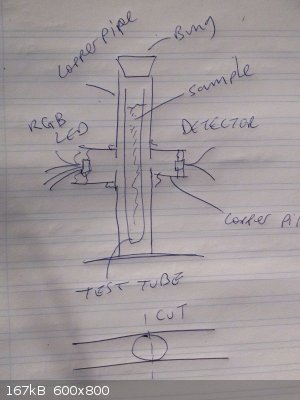
The bottom bit is just an idea of how to make two side-arms fit nicely.
[Edited on 28-5-2015 by aga]
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Smaerd:
With all due respect, the R<sup>2</sup> means very little w/o calculating confidence levels (P values), for estimated (unknown samples)
values.
ALL colourimeters I've ever used use square cuvettes and dark cells. Why change a winning horse? 
[Edited on 28-5-2015 by blogfast25]
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by aga  | A picture speaks a 1000 words
The bottom bit is just an idea of how to make two side-arms fit nicely.
|
Smaller, SQUARE cuvette. Samples cost money. A few ml is better than a few more ml.
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
Still got no square glass tubes, and i've been waiting MINUTES !
Ebay is really letting standards slip.
I can't see how it will matter given the age and dirtyness of my copper pipe - the internal walls are pretty light-absorbent.
As i said, the electronics are the same no matter what setup is used, so may as well try it and see.
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
I suppose lenses and focusing on a capillary tube of sample is being a bit extreme ?
What am i thinking !
A simple reflected light colorimeter would work, and also tell you your skin tone.
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Igor, you drive me into an early grave! Never will I employ an Dago again! 
I will send you one of mine. Maybe Dos if you play nice.
[Edited on 28-5-2015 by blogfast25]
|
|
|
smaerd
International Hazard
    
Posts: 1262
Registered: 23-1-2010
Member Is Offline
Mood: hmm...
|
|
Yea, like I said, this was the simplest colorimeter design I had ever found. It doesn't factor in error associated with multimeter readings, or any of
that. I've also never seen someone calculate a P value for a beers-law plot. Most people just use the error associated with their solution preparation
I thought. Sure a square cuvette is ideal, but like I said, I've used UV-VIS spectrophotometers that used cylindrical cuvettes without any concerns
whatsoever. That's the nice thing about relative measurements.
@aga - now that makes way more sense than what I was picturing lol. Did you ever figure out the linearity of those photocells? If I had a
microcontroller that wasn't being used for a project I was working on, I'd probably join you, but I really think amplification would be more
fun/valuable. Maybe I'll take a crack at amplifying the signal with a basic JFET op-amp later on. I'm excited to see what you come up with though. Do
a beers-law plot with your results when you get them!
Edit - reflected light would work, but you need to factor in that it's now twice the path-length and make sure it's not reflecting awkwardly through
your sample other-wise you will have an unknown path-length.
[Edited on 28-5-2015 by smaerd]
|
|
|
blogfast25
International Hazard
    
Posts: 10562
Registered: 3-2-2008
Location: Neverland
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by smaerd  | I've also never seen someone calculate a P value for a beers-law plot. Most people just use the error associated with their solution preparation I
thought.
|
A lot do, yes. Just don't try and get into 'Nature' with that! 
Believe me that high values of R<sup>2</sup> are deceptive, though.
[Edited on 28-5-2015 by blogfast25]
|
|
|
| Pages:
1
2
3 |