madmaxman
Harmless

Posts: 3
Registered: 12-4-2018
Member Is Offline
Mood: No Mood
|
|
Indoles forming salts?
Can anyone help me understand indole salts?
If you look at tryptophan.hcl it is a salt but due to the primary amine.
If you look at tryptophol, there is no mention of the salt forming.
Is it possible to form a 'double salt' by adding HCL to the indole and amine group?
Can any indole group with a minimum pka form HCL salts? Would that require anhydrous hydrogen chloride or just regular HCL?
I just can't find much information about indole salts, if there was a good textbook or reference on the subject that would be great.
|
|
|
AvBaeyer
National Hazard
   
Posts: 644
Registered: 25-2-2014
Location: CA
Member Is Offline
Mood: No Mood
|
|
The indole ring does not form any useful salts. The nitrogen is simply not basic. You should consult a heterocyclic chemistry text to expand your
knowledge.
AvB
|
|
|
PhenethylamineMachine
Hazard to Others
  
Posts: 110
Registered: 22-3-2018
Member Is Offline
Mood: No Mood
|
|
Hmmm...
I think I understand what you are saying.
You are asking why the NH nitrogen of the indole ring moiety of tryptamine compounds can't also be used to accept a hydrogen ion (H+), as the amine
nitrogen of the compounds does, right?
There is good reason for this, indole is not basic. Because of the aromatic ring the electron pair on that nitrogen are not available for protonation.
I mean, you can protonate indole with certain strong acids, but you get what I am saying.
|
|
|
LearnedAmateur
National Hazard
   
Posts: 513
Registered: 30-3-2017
Location: Somewhere in the UK
Member Is Offline
Mood: Free Radical
|
|
Of course there are strong enough acids, I mean fluoroantimonic acid can create salts from hydrocarbons but common acids just aren’t enough due to
resonance stability.
In chemistry, sometimes the solution is the problem.
It’s been a while, but I’m not dead! Updated 7/1/2020. Shout out to Aga, we got along well.
|
|
|
PhenethylamineMachine
Hazard to Others
  
Posts: 110
Registered: 22-3-2018
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by LearnedAmateur  | | Of course there are strong enough acids, I mean fluoroantimonic acid can create salts from hydrocarbons but common acids just aren’t enough due to
resonance stability. |
Yup, that's what I told the poster, that you can protonate indole with certain strong acids.
However, by context it seemed to me that this was not what the initial poster was asking about.
|
|
|
LearnedAmateur
National Hazard
   
Posts: 513
Registered: 30-3-2017
Location: Somewhere in the UK
Member Is Offline
Mood: Free Radical
|
|
Specifically about HCl, but that’s pretty much the strongest acid one could find in a domestic setting, although I wonder if hydrobromic or
hydroiodic acids could be strong enough as laboratory reagents; doubtful, but there has to be some pKa value which enables acidic protonation at the
nitrogen atom.
In chemistry, sometimes the solution is the problem.
It’s been a while, but I’m not dead! Updated 7/1/2020. Shout out to Aga, we got along well.
|
|
|
madmaxman
Harmless

Posts: 3
Registered: 12-4-2018
Member Is Offline
Mood: No Mood
|
|
Thank you very much, this clears up a lot of confusion for me.
Other compounds similar to this, such as Lysergic Acid Diethylamide, form salts which I thought was at the indole position.
In the case of LSD, is the salt formed at the methylated N position instead?
|
|
|
PhenethylamineMachine
Hazard to Others
  
Posts: 110
Registered: 22-3-2018
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by madmaxman  | Thank you very much, this clears up a lot of confusion for me.
Other compounds similar to this, such as Lysergic Acid Diethylamide, form salts which I thought was at the indole position.
In the case of LSD, is the salt formed at the methylated N position instead? |
Yes, the methylated nitrogen 6 position is the protonation site...primarily. ...it's a actually pretty complicated, but basically N6 is the
protonation site, most of the time.
Below is some review, so you can skip past if needed.
Basics:
·An acid readily gives up a proton (H+)
·A base readily accepts a proton
The conjugate acid is the protonated base, and the conjugate base is what's left of the acid after its donated it's hydrogen ion (proton)
Its pretty cool, if you have the non-protonated from of an amine, the free-base, (called as such because it's "free" from protonation) you know that
an acid/base neutralization will produce a salt, also that the acid used determines the salt produced, so hydrochloric acid gives the hydrochloride
salt, fumeric acid gives the fumarate, acetic acid gives the acetate salt and so on. Each salt form causes the molecule to differ in size, weight,
solubility, and so on. I'm sure this is all review, actually I know it's review based in the conversation so far, it's cool stuff though.
|
|
|
PhenethylamineMachine
Hazard to Others
  
Posts: 110
Registered: 22-3-2018
Member Is Offline
Mood: No Mood
|
|
Wow, I remember there being a related question on a Dartmouth chemistry exam:
In the attached depiction we see that on the test "N2" is representing the methylated nitrogen at position 6 on the LSD molecule.
Ok, here is how this relates to this thread: As the methylated nitrogen at position 6 is the most basic nitrogen atom it would be the most obvious
site for protonation, which again, is a little more complicated, but the information and the link from my last post should have that covered.
Really fascinating stuff. I almost forget that I know some of these things until someone asks about them, then the memories all come rushing back,
coupled with the enthusiasm that was present while first learning these topics. Its been fun responding to this one.
|
|
|
LearnedAmateur
National Hazard
   
Posts: 513
Registered: 30-3-2017
Location: Somewhere in the UK
Member Is Offline
Mood: Free Radical
|
|
Interesting; slightly off topic but does the same hybridisation of the amide group (and amides in general like APAP and acetamide) mean that it is
very difficult to protonate them hence a general inability to form salts? I’m only really familiar with amines, even then just the linear ones as
opposed to heterocyclic compounds, plus I’ve never really gone too far into hybridisation back when I did OC in school - really just covered sp2
with alkenes.
In chemistry, sometimes the solution is the problem.
It’s been a while, but I’m not dead! Updated 7/1/2020. Shout out to Aga, we got along well.
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
"The hybridization of the three nitrogen atoms are N1: sp2; N2: sp3; N3: sp2:
It would be fascinating just to know how to work that out.
[Edited on 16-4-2018 by aga]
|
|
|
Sigmatropic
Hazard to Others
  
Posts: 307
Registered: 29-1-2017
Member Is Offline
Mood: No Mood
|
|
N1 is an amide, the other main resonance structure of an amide has a N=C bond. The nitrogen lone pair is thus said to be delocalized and has full sp2
hydrbridization in that resonance structure. Real world consequences are that this nitrogen is non basic, rotation about C-N bond is restricted and
the N is trigonal planar.
N2 is a normal amine, it has a sp3 hybridization typical of 2nd row elements. This N is basic and trigonal pyrimidal.
N3 is part of a hetereoaromatic ring (see Huckel's rule for aromaticity). The nitrogen lone pair is needed to fufill Huckel's rule and can be
considered to be delocalied into the ring. This nitrogen is non basic and trigonal planar.
Now for a bonus, what are the most acidic protons in LSD and their relative order?
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
Cool ! Thanks for that explanation.
Eeek! That's a hard one.
Umm, 'acidic' meaning more H-plussy isn't it ?
If so, electronegativity should come into it, so my guess:
The H on N3
The C next to N1
No idea really.
It'd be great to know how to work that out too.
|
|
|
Sigmatropic
Hazard to Others
  
Posts: 307
Registered: 29-1-2017
Member Is Offline
Mood: No Mood
|
|
It is indeed the H on N3 but this indolic NH can only be deprotonated by strong bases. Although I did indeed mean this proton when I said protonS, I
doubt it can actually be deprotonated in ergolines (which is, I think the result of the acidic protons elsewhere on the molecule. What I mean is I
don't think there will be a double anion).
It is not the C next to the N1 but very close by alpha protons of the amide. Deprotonation at that position gives quite a large conjugated system and
is actually the basis of the isomerization between iso-LSD and LSD.
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
Holy crap !
I guessed right !
So electronegativity is part of how you work that kind of stuff out ?
OC is still Dark Magic to me.
|
|
|
Sigmatropic
Hazard to Others
  
Posts: 307
Registered: 29-1-2017
Member Is Offline
Mood: No Mood
|
|
It is all guided by pKa values which are inherently connected with the stability of the conjugate bases. This stability is indeed a function of
electronegativity but also other factors such as size (1), inductance (2) and resonance (3).
(1) Take for example the difference between the pKa of an alcohol and a thiol. On first inspection Oxygen is more electronegative and hence should be
more 'willing' to accept electrons. This would lead to the wrong conclusion that alcohols are more acidic than thiols. Sulfur is a third row element
and it's valence electrons are in a larger orbital, this results in a more spread out charge and is thus more stable.
(2) effect created by the presence of nearby, but not connected electronegative groups. For example compare trifluoroacetic acid, trichloroacetic acid
and acetic acid.
(3) caused by the presence of connected electron withdrawing groups (note the difference between electronegative and electron withdrawing). For
example compare the acidities of p-nitrophenol and m-nitrophenol.
This get's complicated in organic molecules, although it is really more of the same, as one can have multiple acidic protons. Deprotonation at one
site affects the other site (as can clearly be seen the simple case of oxalic acid). A general knowledge of pKa values for various functional groups
is very helpfull at this point. I would say the way to figure this out is by taking into account all these factors and a bit of knowledge. That is
noting that 'This proton is next to an electron withdrawing carbonyl' or 'this proton in on a hetereo atom of a certain type'. Then knowing that amide
bases (LDA, KHMDS, LiHMDS, NaNH2, ect) can be used to deprotonate carbonyl compounds (aldehydes, ketones, esters, amides, nitriles, ect) and the
relative order follows: amide bases are stronger bases than enolates. At best it is performed on intuition and will not necessarily be correct.
|
|
|
aga
Forum Drunkard
    
Posts: 7030
Registered: 25-3-2014
Member Is Offline
|
|
blogfast25 did teach something of what pKa is, yet my addled brain does not recall right now.
pKa is a measure of how acidic a certain thing is, i think.
Great teaching so far, keep it up !
Edit :
PLEASE
[Edited on 16-4-2018 by aga]
|
|
|
LearnedAmateur
National Hazard
   
Posts: 513
Registered: 30-3-2017
Location: Somewhere in the UK
Member Is Offline
Mood: Free Radical
|
|
It’s basically the measure of how an acid dissolves in water - the lower the pKa (it’s a negative logarithm) the more protons are released into
solution. Hence, sulphuric acid releases more protons than acetic acid, and hydrochloric acid releases more than both, as examples. Each decrease by
-1 means that 10 times as many H+ ions are released.
Edit: I may be a bit inebriated.
[Edited on 16-4-2018 by LearnedAmateur]
In chemistry, sometimes the solution is the problem.
It’s been a while, but I’m not dead! Updated 7/1/2020. Shout out to Aga, we got along well.
|
|
|
zed
International Hazard
    
Posts: 2277
Registered: 6-9-2008
Location: Great State of Jefferson, City of Portland
Member Is Offline
Mood: Semi-repentant Sith Lord
|
|
Form a salt at position 1? Not impossible, but in doing so, Indole would cease to be Indole.
Indoles can fussily self-destruct under some conditions.
It is possible by various means, to hydrolyse the enamine area of the ring.... opening it. But, then of course, it isn't an Indole anymore.
Kyanuric acid comes to mind. I'll check.
Well, I checked. Might have the wrong spelling. I searched, but in vain.
[Edited on 17-4-2018 by zed]
|
|
|
Sigmatropic
Hazard to Others
  
Posts: 307
Registered: 29-1-2017
Member Is Offline
Mood: No Mood
|
|
Salts the one position are intermediates in some reactions (methylation, tosylation) and the products are still very much indoles. I don't understand
what you mean with that it would cease to be indole.
|
|
|
LearnedAmateur
National Hazard
   
Posts: 513
Registered: 30-3-2017
Location: Somewhere in the UK
Member Is Offline
Mood: Free Radical
|
|
Wouldn’t it remove the electron delocalisation if you withdrew electrons from the ring during protonation, hence why it would destroy the indole
structure?
In chemistry, sometimes the solution is the problem.
It’s been a while, but I’m not dead! Updated 7/1/2020. Shout out to Aga, we got along well.
|
|
|
PhenethylamineMachine
Hazard to Others
  
Posts: 110
Registered: 22-3-2018
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by LearnedAmateur  | | Wouldn’t it remove the electron delocalisation if you withdrew electrons from the ring during protonation, hence why it would destroy the indole
structure? |
Electrons are not withdrawn during protonation.
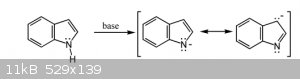
|
|
|
LearnedAmateur
National Hazard
   
Posts: 513
Registered: 30-3-2017
Location: Somewhere in the UK
Member Is Offline
Mood: Free Radical
|
|
Alright, thanks for that, wasn’t sure if the lone pair was donated to the ring or not. Quite a helpful diagram.
In chemistry, sometimes the solution is the problem.
It’s been a while, but I’m not dead! Updated 7/1/2020. Shout out to Aga, we got along well.
|
|
|
byko3y
National Hazard
   
Posts: 721
Registered: 16-3-2015
Member Is Offline
Mood: dooM
|
|
https://books.google.com/books?id=srxzzUskq4wC&pg=PA51
pKb of unsubstituted protonated indole is -4 (pKb = 18), thus this acid is stronger than sulfuric acid. It will be formed under special conditions,
but that stuff is very unstable.
2-Methylindole hydrochloride is sold commercially though, just like dimethylformamide hydrochloride and other stuff with pKb above 14.
|
|
|
PhenethylamineMachine
Hazard to Others
  
Posts: 110
Registered: 22-3-2018
Member Is Offline
Mood: No Mood
|
|
I actually had some long highly detailed post discussing protonation, deprotonation, and discussion revolving around that non-bonding pair, but during
editing a glitch in my device caused me to loose the post, so I quickly typed out a single sentence and posted. ...however, the picture I uploaded was
still there.
I am glad it helped, but apologize that the actual post was destroyed, but if you got what you needed from it I guess it all worked out.
|
|
|