
amrhamed2 - 21-3-2008 at 10:42
The idea is to convert 1 to 2 then followed by facile decarboxylation
There is a method stated in the literature for this conversion .
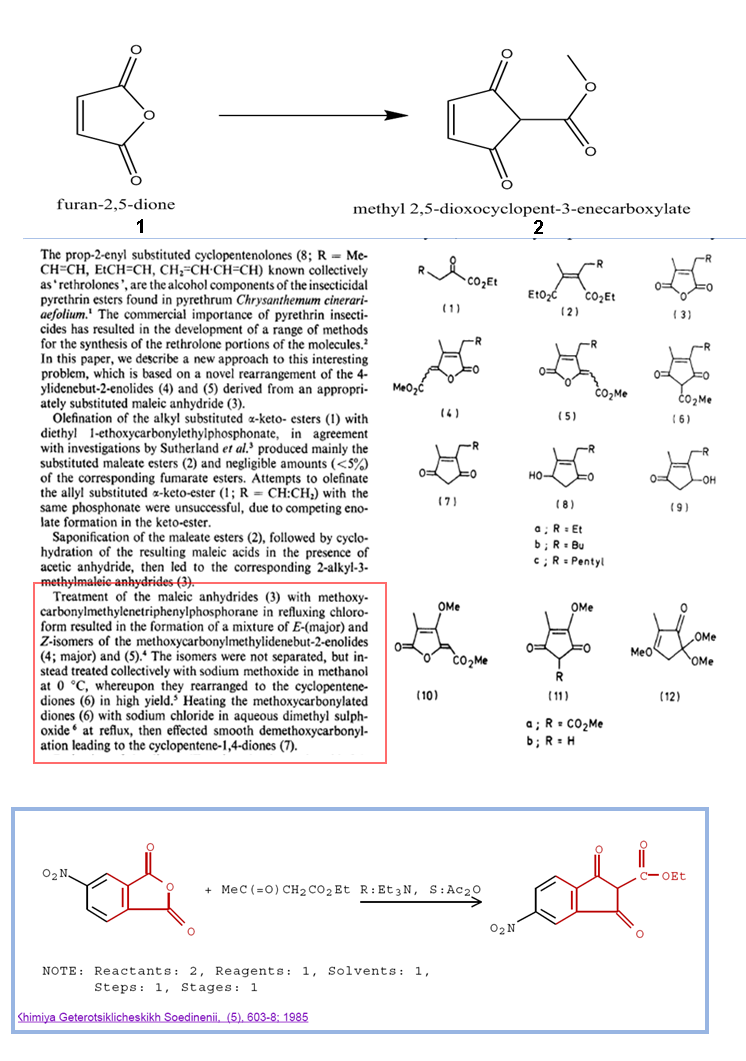
It is illustrated in the image below (in the middle)using Ph3 PCH = COOMe (methoxycarbonylmethylenetriphenylphosphorane)
I wonder if I can use the method in khimiya journal (the one in the bottom) using methylacetoacetate and triethylamine followed by decarboxylation
[Edited on 21-3-2008 by amrhamed2]
[Edited on 21-3-2008 by amrhamed2]
amrhamed2 - 21-3-2008 at 13:06
 Can anyone explain what happened in the reaction stated in khimiya journal and
tell me whether it will work for maleic acid or not
Can anyone explain what happened in the reaction stated in khimiya journal and
tell me whether it will work for maleic acid or not
Dr. Beaker - 21-3-2008 at 15:53
In 1st glance it looks like it involves aldol condensation.
btw - does anyone cite this paper
chemrox - 21-3-2008 at 23:35
would you please type out the cite and possibly post the paper since you obviously have it? It's considered bad form to take up a whole page in the
thread with a copy of a page from a paper instead of sharing the cite. it's also considered rude to ask questions based on a paper without citing the
paper.
[Edited on 21-3-2008 by chemrox]
kmno4 - 22-3-2008 at 01:43
This "khimiya journal" is available from Springer as "Chemistry of heterocyclic compounds" (translated Khimiya
Geterotsiklicheskikh Soedinenii). Also available from "Full volumes..." thread.
Article is from volume 21, p. 501-506.
amrhamed2 - 22-3-2008 at 12:16
first article
J. CHEM. SOC. PERKIN TRANS. I 1983
This is the one in the middle
Attachment: P19830000467.pdf (319kB)
This file has been downloaded 741 times
amrhamed2 - 22-3-2008 at 12:40
Something strange in the second article...........
I read the translation ..........it deals with indanes and not the structure found in the scifinder search .........
here is the article
Attachment: fulltext.pdf (393kB)
This file has been downloaded 714 times
amrhamed2 - 28-3-2008 at 00:10
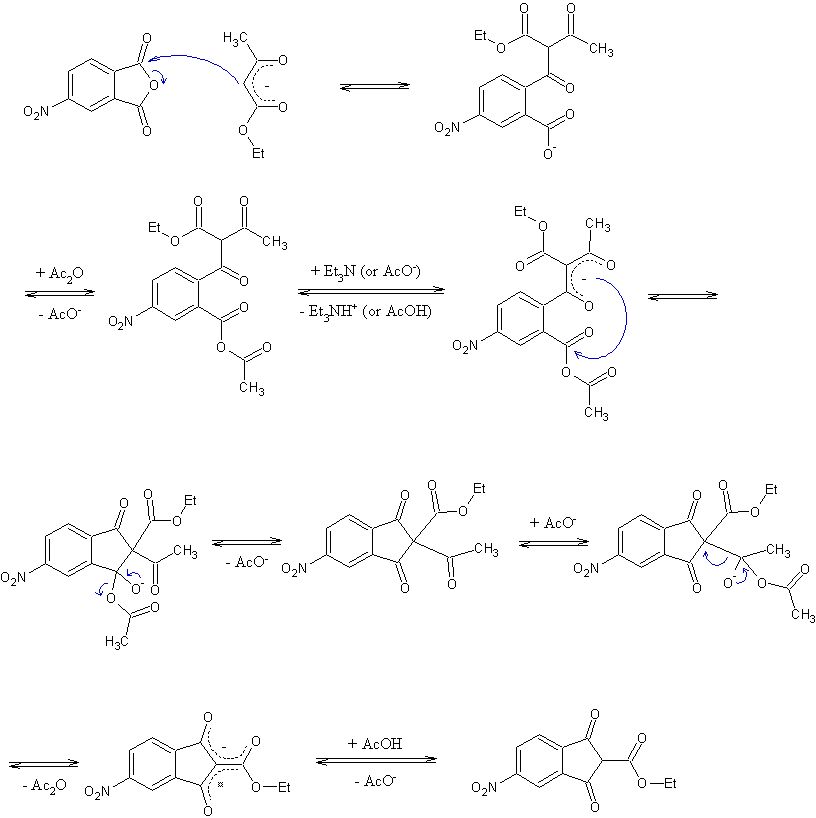
Can anyone explain to me the mechanism of reaction between methylacetoacetate and cyclic anhydride mentioned in khimiya journal .....
Nicodem - 28-3-2008 at 02:45
I really hope your request has a purpose as it took me almost half an hour to draw all the damn arrows. Besides as a graduated chemist (I guess you
finished school already given you do research) you are expected to know how to draw mechanisms, especially simple ones like acyl condensations and
similar ones involving only electron pair transfers.
amrhamed2 - 28-3-2008 at 05:12
I am really thankful Nicoderm for helping me .I really appreciate it .
In fact I didn't study mechanistic organic chemistry in depth ,and i would like you to recommend me some books to read in mechanistic and physical
organic chemistry . I know March and Carey advanced organic chemistry ,but I need some simpler approaches (crash like course)
Nicodem - 28-3-2008 at 06:56
For a complete beginner who wants to know how to write reasonable organic reaction mechanisms, I would advise this book:
The Art of Writing Reasonable Organic Reaction Mechanisms by Robert B. Grossman (2003, ISBN: 978-0-387-95468-4)
It is certainly the most comprehensive book for those who want to learn how to "push arrows".
PS: It's "Nicodem", not "Nicoderm".
[Edited on 28/3/2008 by Nicodem]
amrhamed2 - 28-3-2008 at 07:06
Here is a a link to the book h##p://rapidshare.com/files/103046154/art.of.writing.reasonable.organic.reaction.mechanisms-0387954686.rar
Thanks Nicoderm

 Can anyone explain what happened in the reaction stated in khimiya journal and
tell me whether it will work for maleic acid or not
Can anyone explain what happened in the reaction stated in khimiya journal and
tell me whether it will work for maleic acid or not