
amrhamed2 - 12-11-2008 at 08:10
In order to substitute the chlorine with different anilines the author used acidulated water or benzene
I don't have the paper so I don't know the details but from my experience I know that this ester is very reactive and can form amide with amines
easily
I am just wondering .........does this reaction proceeds at rt or needs reflux (step 2) and how to avoid reaction between ester and amine if reflux
was used
[Edited on 12-11-2008 by amrhamed2]
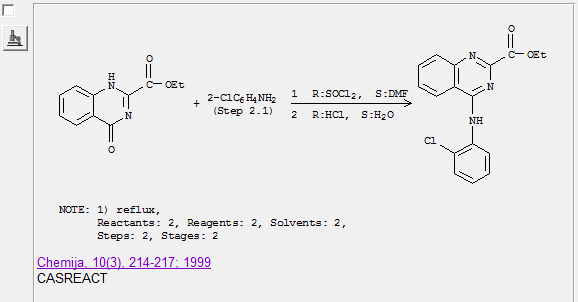
amrhamed2 - 12-11-2008 at 08:12
Here is the abstract of the paper
Synthesis and antiinflammatory activity of ethyl 4-(anilino substituted)-2-quinazolinecarboxylates. Mekuskiene, Giedrute; Udrenaite, Emilija;
Gaidelis, Povilas; Vainilavicius, Povilas. Faculty of Chemistry, Vilnius University, Vilnius, Lithuania. Chemija (1999), 10(3), 214-217.
Publisher: Academia, CODEN: CHMJES ISSN: 0235-7216. Journal written in English. CAN 133:362749 AN 2000:675531 CAPLUS
Abstract
Treatment of Et 4-oxo-3,4-dihydro-2-quinazolinecarboxylate with thionyl chloride resulted in Et 4-chloro-3,4-dihydro-2-quinazolinecarboxylate
formation. The latter reacted with arom. amines in acidified water or benzene to form Et 4-(anilino substituted)-2-quinazolinecarboxylate possessing
antiinflammatory activity.
Klute - 13-11-2008 at 05:51
Aminolysis of desactivated esters with aromatic amines requires very hot temperatures and oftene xcessive reaction times.
When adding SOCl2 to the substarte, youa re forming a chloro-iminium species, which is very reactive and will attack the amine long before the ester.
So no worry on that side IMHO.
