Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
FNG here, how can I contribute to the energetics forum.
Hello. I'm new. I’ve been referencing SM for about five years, But have only just now made an account. I am still new to Chemistry, but have been
unknowingly doing it since a young age. I have professional field experience with demolitions, energetic compositions, techniques and procedures. Is
there any particular area of energetics that I could contribute to that would be useful or of interest to members? I have blast pits and impact
ranges.
|
|
|
B(a)P
International Hazard
    
Posts: 1114
Registered: 29-9-2019
Member Is Offline
Mood: Festive
|
|
Welcome to the forum! Sounds like you have a pretty amazing set up.
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Thank you for the warm welcome. I agree, the earth is incredible and amazing. Is there any area of interest to you that has been difficult to validate
or find needed insight into? I would like to focus research and documentation on those if possible. My personal interest is pbx, rocket and ballistic
propulsion, mechatronics, pneumatics, metals, cellulose and electrolysis, energy, pyrolysis, radio, lasers and agriculture. I would like to focus
effort on areas that would be helpful to others on the forums. If possible. --I'm quite amateur and not very smart, but I have a lot of practical
experience with demolition ranging from hmx to random chemical explosions in universities, im interested in addressing needed areas and welcome
suggestions to begin research.
[Edited on 3-11-2020 by Hey Buddy]
|
|
|
B(a)P
International Hazard
    
Posts: 1114
Registered: 29-9-2019
Member Is Offline
Mood: Festive
|
|
My professional life frequently merges into the world of energetics so I am quite lucky in that I have both the qualifications and space available to
experiment with about as much as I want to.
Don't sell yourself short, people that are not smart don't last particularly long in the energetics industry . I am also an amateur and have always been fascinated by chemistry. . I am also an amateur and have always been fascinated by chemistry.
I look forward to seeing your contributions, I am sure there are many here that will take you up on your offer.
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
I really hope I don't let the forum down. I'd like to ask you a personal question if you don't mind. What would you do with a few hundred idle kg of
this? I'm leaning towards separation and recomposition to more useful synthesis stocks. But I'm not sure what sort of products it can be expediently
or sanely rendered into. Sodium Nitrate > Sodium Nitrite + Sodium Chloroacetate> Nitromethane is one consideration. Or combination with
Potassium Sulate > Potassium Nitrate. I dont know what use the lye would have other than making some soap. I also haven't figured out how to
separate. I'm assuming just heating in water? Idk. I just got a lot of chemicals disposed onto my care about 10 minutes ago...
--Tough crowd... drinks on me. just finished loading up all these chemicals from a municipal disposal. Several hundred quarts of unopened industrial
non-commercial H2SO4 drain cleaner. If anyone wants some H2SO4 in the area, you can have some. Im in western Missouri near I70.

[Edited on 4-11-2020 by Hey Buddy]
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Well, I guess ther're no sciencey ppl in this area at this time so unfortunately I'm going to have to put all this H2SO4 in longterm glass and it
won't be convenient to give away at that point. I tried. Probably a bad night, lots of "other" stuff going on. I will probably make NM and Nitric
stock from all this later. Any suggestion for separating the lye and sodium nitrate? It would be much appreciated. Ive ever done that.

|
|
|
Tsjerk
International Hazard
    
Posts: 3022
Registered: 20-4-2005
Location: Netherlands
Member Is Offline
Mood: Mood
|
|
The only way I can think of to separate lye and sodium nitrate is by adding ammonium nitrate. You would have to dissolve everything and boil off the
ammonia while boiling of the water. That way you don't actually seperate the two but you turn the lye into sodium nitrate.
|
|
|
B(a)P
International Hazard
    
Posts: 1114
Registered: 29-9-2019
Member Is Offline
Mood: Festive
|
|
Quote: Originally posted by Hey Buddy  | Well, I guess ther're no sciencey ppl in this area at this time so unfortunately I'm going to have to put all this H2SO4 in longterm glass and it
won't be convenient to give away at that point. I tried. Probably a bad night, lots of "other" stuff going on. I will probably make NM and Nitric
stock from all this later. Any suggestion for separating the lye and sodium nitrate? It would be much appreciated. Ive ever done that.
|
If you mean by 'other stuff' the state of Australian rugby league State of Origin and by in this area you mean Sydney, Australia I am happy to forgo
the football and come over and take some of that off your hands. 
edit - Sorry, missed your location in the earlier post
[Edited on 4-11-2020 by B(a)P]
|
|
|
Laboratory of Liptakov
International Hazard
    
Posts: 1335
Registered: 2-9-2014
Location: Technion Haifa
Member Is Offline
Mood: cool.gif
|
|
According to series (and contradictory content) of quick comments and photos, it looks like the chemicals seized in the police warehouse .... And you
are neweler to the warehouse. And no one has told you, a what to do with it .....Therefor you are here......
[Edited on 4-11-2020 by Laboratory of Liptakov]
Development of primarily - secondary substances CHP (2015) Lithex (2022) Brightelite (2023) Nitrocelite (2024)
|
|
|
j_sum1
Administrator
       
Posts: 6220
Registered: 4-10-2014
Location: Unmoved
Member Is Offline
Mood: Organised
|
|
Quote: Originally posted by Laboratory of Liptakov  | According to series (and contradictory content) of quick comments and photos, it looks like the chemicals seized in the police warehouse .... And you
are neweler to the warehouse. And no one has told you, a what to do with it .....Therefor you are here......
[Edited on 4-11-2020 by Laboratory of Liptakov] |
LL! Lovely to see you back – it has been a while.
There have been some concerns as to whether you were still ok.
|
|
|
Laboratory of Liptakov
International Hazard
    
Posts: 1335
Registered: 2-9-2014
Location: Technion Haifa
Member Is Offline
Mood: cool.gif
|
|
Hi, Sum..... .....I am all right....Somehow I missed the theories (sometimes
nonsense) that come up here.... .....I am all right....Somehow I missed the theories (sometimes
nonsense) that come up here....
I'm just beating a coronavirus on the Anvil now...With a big hammer...
Now is he almost flat. I guess....
[Edited on 4-11-2020 by Laboratory of Liptakov]
Development of primarily - secondary substances CHP (2015) Lithex (2022) Brightelite (2023) Nitrocelite (2024)
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Quote: Originally posted by Tsjerk  | | The only way I can think of to separate lye and sodium nitrate is by adding ammonium nitrate. You would have to dissolve everything and boil off the
ammonia while boiling of the water. That way you don't actually seperate the two but you turn the lye into sodium nitrate. |
I really do appreciate this.
|
|
|
Herr Haber
International Hazard
    
Posts: 1236
Registered: 29-1-2016
Member Is Offline
Mood: No Mood
|
|
Great idea Tsjerk !
Funny someone else is trying to separate another drain opener with a similar formula in another thread.
Buddy, all that H2SO4 while there is a ban in Europe will make people cry. Bad Buddy !
The spirit of adventure was upon me. Having nitric acid and copper, I had only to learn what the words 'act upon' meant. - Ira Remsen
|
|
|
Hey Buddy
Hazard to Others
  
Posts: 384
Registered: 3-11-2020
Location: Bushwhacker Country
Member Is Offline
|
|
Following up on this. Particularly drain cleaner component separation. It seems to be a common issue. People buying drain cleaner and discovering it
contains NaOH, NaNO3, Al, and a dye. In this case, I was interested in recovery of components due to the amount, which was bulk. But i wouldn't
recommend buying the mixed product with the intent to separate.
Out of curiosity, I added .5L of the mixed drain opener to a stainless pot with 1L of water @~20C. Mixture began bubbling and steaming within 45
seconds and proceeded to foam and off gas heavily with overflowing red dye/lye foam reaching a temp of 100c. Very caustic fumes.
For separation, I found an aluminum screen to work well to pass the NaOH through and retain the Al and NaNO3. Most of the dye passes through with the
NaOH on screening. This should be done outside. I tried a sheet of Al screen door material to filter the material at first but it was very slow and
unwieldy so I found using a section of pvc pipe with screen taped across one end and the other side capped to be fastest. Then I filled up the tube,
capped it and shook it over a bucket. This left NaNO3 and Al in the tube with residual dye.
The remaining risidual dye on the NaNO3 can be removed by a short cold water bath, leaving white NaNO3 prills and Al after the cold water is poured
off.
Then the NaNO3 is heated to dissolve in water, Al separated by filter. recrystalized by boiling. Then filtered to remove remaining dye and can be
cleaned further with acetone wash if necessary.
For drying NaNO3, I used a sheet of glass with nitrate spread across as a thin paste. When it became brittle, it was scraped into a glass jar and a
piece of PTFE gas tape was stretched over the mouth and taped secure, allowing moisture to escape. I put the jar in front of a heater to accelerate
drying.
 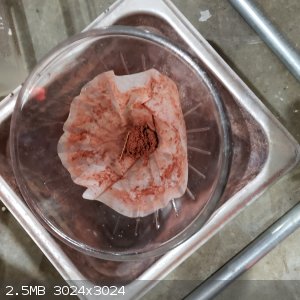 
[Edited on 4-1-2021 by Hey Buddy]
|
|
|
roXefeller
Hazard to Others
  
Posts: 463
Registered: 9-9-2013
Location: 13 Colonies
Member Is Offline
Mood: 220 221 whatever it takes
|
|
Spheroidization of DDNP? Rosco and Hennig Brand were working on something like this but it seemed like an elusive grail from chinese researchers.
One must forego the self to attain total spiritual creaminess and avoid the chewy chunks of degradation.
|
|
|