Morgan
International Hazard
    
Posts: 1750
Registered: 28-12-2010
Member Is Offline
Mood: No Mood
|
|
Sensitizing Nitrogen Triiodide
So the other day I was attempting to put a small amount of iodine crystals into this long quartz tube.
https://www.sciencemadness.org/whisper/files.php?pid=692476&...
https://www.sciencemadness.org/whisper/files.php?pid=692476&...
The original bottle and degrading cap I had bought iodine in long ago was starting to fail after a few years, so I had put it in a small cobalt blue
bottle to which I wrapped some Teflon tape around the threads to slow the sublimation and then stored it that way. The crystals didn't want to come
out so I used a glass rod to breaks some crystals free but when I went to sprinkle some too much came out of the hard to see inside bottle and I
didn't feel like putting the excess back in the bottle.
Being bored I was going to see how an iodine vapor cloud would look when heated in the long glass tube. After doing that and not all that exciting, I
decided to make some triiodide using some household ammonia. But when the hydroxide mixed with the iodine I couldn't tell how much was reacting
because the previously melted iodine solidified into a solid mass at the bottom of the quartz tube. I took a long flimsy wooden dowel to try to break
up the rock-solid fused iodine lump stuck in the bottom of the quartz tube.
After filtering what triiodide would come out onto filter paper a hard remaining mass still wouldn't dislodge and leaving it in the quartz tube made
me nervous. I filled the tube with water and still tried to crunch the mass apart with a thin wooden dowel but no luck.
That is when I decided to try some methanol to dissolve it out that way. It dissolved some but not that much and I noticed a few minute pops and it
strangely seemed if they were happening under the methanol, a thunking sound.
The next day I went in the yard where the tube was left to sublimate away and looking down into the tube a violet vapor could be seen. So still being
too slow to rid it of the tube, I heated the mass and it solidified into two long streaks/lines as I tried to pour the liquid iodine out. I left that
out another day and still it was hardly gone with the greater surface area exposed. Bothered by the curious pops initially, I started to wonder if
methanol had sensitized the triiodide so again I filled the tube with household ammonia whilst rubbing the sides of the tube to dislodge the iodine
with a dowel. The whole tube eventually turned a dark black and most of the solids were poured out onto filter paper. The crumbs that were left in the
tube then had about 100 ml of methanol poured into the tube for the test.
An occasional popping sound occurred as the wooden dowel was swished around in the solution or if the tube was jiggled. But still I couldn't be sure
the thunks were happening under the methanol or just on the wet walls above the solution. Next, some of the triiodide was smudged onto the end of the
flimsy wooden dowel and carefully lowered into the methanol at the bottom of the tube which was held partly sideways. As soon as the sludge entered
the solution or sometimes a few seconds after, a modest "underwater" explosion occurred. Several more trials of this proved positive that that was
what was happening. On one occasion the solution "bumped" quite violently which made me somewhat nervous, the sound and vibration to my hand was quite
alarming as a potentially shattering glass tube isn't fun to be around.
In an old college chemistry demonstration manual, there was mention of one rare occurrence of triiodide exploding while wet. I'm pretty sure now
methanol would be another way to test the hypothesis.
The experiment brought to mind the ethanol, sulfuric acid, and KMNO4 reaction for some reason, though that more subdued.
|
|
|
Morgan
International Hazard
    
Posts: 1750
Registered: 28-12-2010
Member Is Offline
Mood: No Mood
|
|
Some afterthoughts about this methanol triiodide experiment ...
I think on a small scale this could be an interesting demonstration as the triiodide explodes under the fluid methanol when stirred or the vessel
jiggled. I wonder if methanol dries the triiodide in a sense or if the pH is doing something to create the interesting pops and explosions without any
sign of the typical triiodide smoke of iodine as it explodes under the fluid? The methanol used was a product called HEET, when you have water in your
gasoline.
It's a very strange sensation, a dark black solution making random thunking sounds when agitated or stirred. If you had felt some of the stronger
"pings" I felt, it starts to get exciting in a risky way.
This is the permanganate experiment I referenced in the initial account.
Lightning in test tube : reaction of Potassium Permanganate and Sulphuric acid
https://youtu.be/Rt5BToe-JDw?t=161
|
|
|
Radiums Lab
Hazard to Others
  
Posts: 299
Registered: 18-3-2025
Location: India
Member Is Offline
Mood: Experiencing the elegance of science.
|
|
Use NaOH sol. to recover and neutralise leftover I2. NaI is also a precious reagent.
[Edited on 12-11-2025 by Radiums Lab]
Water is dangerous if you don't know how to handle it, elemental fluorine (F₂) on the other hand is pretty tame if you know what you are doing.
|
|
|
Radiums Lab
Hazard to Others
  
Posts: 299
Registered: 18-3-2025
Location: India
Member Is Offline
Mood: Experiencing the elegance of science.
|
|
But I don't know aq. NaOH's reactivity with NI3.
Water is dangerous if you don't know how to handle it, elemental fluorine (F₂) on the other hand is pretty tame if you know what you are doing.
|
|
|
DraconicAcid
International Hazard
    
Posts: 4503
Registered: 1-2-2013
Location: The tiniest college campus ever....
Member Is Offline
Mood: Semi-victorious.
|
|
I can't give you a source, but I'm pretty sure it hydrolyzes it to nitrite ion and iodide.
Please remember: "Filtrate" is not a verb.
Write up your lab reports the way your instructor wants them, not the way your ex-instructor wants them.
|
|
|
Radiums Lab
Hazard to Others
  
Posts: 299
Registered: 18-3-2025
Location: India
Member Is Offline
Mood: Experiencing the elegance of science.
|
|
So are we getting NaNO2? That's intresting for fellow US members.
Water is dangerous if you don't know how to handle it, elemental fluorine (F₂) on the other hand is pretty tame if you know what you are doing.
|
|
|
Morgan
International Hazard
    
Posts: 1750
Registered: 28-12-2010
Member Is Offline
Mood: No Mood
|
|
I wonder too with the snappy triiodide explosions in the inky black fluid methanol, if some cavitation could be adding to the strange sharp pinging
effect felt in the fused quartz tube when holding it? Whatever the case, it's quite wondrous, fun, and suspenseful. You know you are playing with
energy.
An aside,
The same quartz tube repurposed for another experiment.
Decibel Level Trial with a Quartz Tube
https://www.youtube.com/watch?v=4mcFjwwDV84
Peak dB Experiment with a Quartz Tube
https://www.youtube.com/watch?v=si8EuIR0GO8
How Loud Is Opening An Aluminum Drink Can?
https://www.youtube.com/watch?v=w9zdlMRgTjw
After rereading some posts, I came across this account. While not like my experiment, completely submerged in methanol dampening the explosions, just
rinsing with water is apparently enough.
Quote: Originally posted by spotlightman1234
You never want to wash NI3 with water or any other solvent!! nitrogen triiodide forms an adduct with ammonia
NI3-NH3-NI3-NH3.... this is why the nitrogen triodide is stable when wet in ammonia, but when it is allowed to dry the ammonia leaves, leaving only
NI3.. I tested this by (from a distance of course) washing wet NI3 with water to get rid of the ammonia. After about 200ml of water were washed
through the filter paper some of the NI3 exploded, spreading NI3 everywhere making a fun residual crackling sound.
[Edited on 13-11-2025 by Morgan]
|
|
|
Radiums Lab
Hazard to Others
  
Posts: 299
Registered: 18-3-2025
Location: India
Member Is Offline
Mood: Experiencing the elegance of science.
|
|
@Morgan so you mean the NI3 will explode on contact with aq. NaOH?
Water is dangerous if you don't know how to handle it, elemental fluorine (F₂) on the other hand is pretty tame if you know what you are doing.
|
|
|
Morgan
International Hazard
    
Posts: 1750
Registered: 28-12-2010
Member Is Offline
Mood: No Mood
|
|
From my experiment I found NI3 will explode submerged in methanol and I referenced a finding or an account where just rinsing with water removes the
ammonia which was reported or needed to stabilize the explosive. I do not know the effect of "aqueous NaOH" as I never used it. But it's a good
question.
What I would like to know is if when I put a small lump of nitrogen triiodide in ~100 ml of methanol, and the long quartz tube kicked strongly,
luckily without shattering, if the substance was diffuse or dissolved through the solution and some sort of cascade occurred or the mushy paste mostly
settled on the bottom and exploded. The solution was a dark black so I couldn't see if it dissolved when I swished it with a long narrow wooden dowel
and jiggled the tube setting it off. Dark black because prior after pouring out most of the newly made triiodide into a coffee filter, I left some
particles/sediment along the length of the quartz tube which got rinsed off and into the methanol when methanol was added for the experiment. In that
way it rinsed off the sides of the tube on the way down to the bottom of the long quartz tube when methanol was added.
Lastly, "if" iodine is happy to take NH3 from a very weak watery ammonium hydroxide solution, and just giving it more time to react, it might be a
risky way to make it. Or then, a supposition, you could make something like what I did and observe attention-getting energetic "thumps" from a liquidy
fluid, in this variation/scenario a watery ammonium hydroxide solution instead of methanol. Or perhaps just dunk some in a tube filled with water and
see what happens instead of rinsing with 200 ml of water on filter paper in the account referenced.
Whatever the case, never make more than you want to blowup in your face. Be very careful and take precautions if attempting.
[Edited on 13-11-2025 by Morgan]
|
|
|
Morgan
International Hazard
    
Posts: 1750
Registered: 28-12-2010
Member Is Offline
Mood: No Mood
|
|
A follow up on sensitized nitrogen triiodide with methanol ... I put less than a teaspoon of iodine in a Tri clover sanitary steel jar/bottle, added
some clear household ammonia, and then slowly decanted the liquid. The muddy triiodide mostly remained in the jar to which about 150 ml or so of water
was added. Next, try as I might, tapping and prodding the bottom of the jar with a meter long thin wooden dowel, I couldn't get any pops or "bumping".
So, all this was disposed of, dried and exploded on filter paper and the test started anew.
The next trial, about a teaspoon of iodine was added to the bottle with about 150 ml of old household ammonia. The crystals were stirred in the steel
bottle tapping and crunching the iodine to break it up and react again as before. With the reaction complete, the liquid portion was slowly
tilted/decanted so that the heavy pasty solids remained near the bottom. Then about 150 ml of methanol was added to the muddy paste at the bottom of
the jar.
All this was done at an angle so that my face or hands weren't directly above the heavy walled steel jar. Prodded lightly with the wooden dowel once
or twice a few energetic pops occurred originating from the submerged explosive. The jar was then moved off the deck and onto a steppingstone. The
very next tap on the bottom of the jar with the flimsy wooden dowel caused an impressive explosion, blasting all the material and methanol straight up
in the air, reminiscent of some trichloride videos seen on YouTube. Although my hand was not directly above the jar and about a meter away holding the
end of the dowel, it got hit with some of the spread or spray and stung for just a brief moment, leaving my hand with an iodine stain. I think having
the methanol at least dampens some of the energy of an otherwise dry triiodide.
On yet another trial, the triiodide exploded without any provocation, quite a surprise, just about a minute after adding the methanol. The blast with
the methanol and submerged iodide is an interesting effect. If you don't like waiting for wet triiodide to dry, this is the way to go.
I asked AI on my phone the reaction of methanol with triiodide and got this funny answer.
Handling in Solution: In a solution like methanol, the compound is more stable and less prone to spontaneous detonation compared to its dry form.
However, reports from amateur chemistry forums indicate that even wet nitrogen triiodide under methanol can produce "underwater explosions" if
disturbed, albeit less sensitive than when dry.
A note of caution, your results may vary. It would be a good idea to wear a face shield and hearing protection. And never look down into a bottle or
put your face or body over or in the path of a possible trajectory of the blast. Just adding methanol to newly made triiodide means danger time has
started the instant the two combine.
Oddly, when I added methanol to an ammonium hydroxide/triiodide solution without decanting the ammonium hydroxide, for some reason most of the
triiodide didn't even pop or snap as it dried on the ground or on filter paper, reverting to iodine passively perhaps.
 
[Edited on 15-11-2025 by Morgan]
|
|
|
Sulaiman
International Hazard
    
Posts: 3938
Registered: 8-2-2015
Member Is Offline
|
|
interesting ......
this is now on my to-do list.
a bend in your poking rod can keep your hand(s) out of the line of fire.
CAUTION : Hobby Chemist, not Professional or even Amateur
|
|
|
Morgan
International Hazard
    
Posts: 1750
Registered: 28-12-2010
Member Is Offline
Mood: No Mood
|
|
Today I tried putting a pinch of iodine in a small test tube and then added some household ammonia and waited for about 20 minutes. Then the tube was
carefully tilted and the hydroxide decanted. The iodide did a pretty good job of staying on the bottom of the test tube without the need to filter
paper or gather it. Next 5 ml of methanol was added, the green silicone extension so that it could be added on the safe side of a -2 cm thick acrylic
picture frame, two magnetic pieces really.
With a long wooden dowel the mixture was tested/gently stirred and only a faint pop occurred under the methanol. Then, becoming more aggressive, as
much as I jabbed at it nothing else happened after that. Recalling a previous experiment where after some time and a partial but big explosion with
the steel bottle previously shown, the tri clover bottle sat with still more fluid remaining and out of the blue it exploded again, this time deciding
to explode without my help. That got my attention.
Anyway, back to the test tube, after leaving it along for 5 minutes, keeping the above account in mind, I again went to stir the solution with the
long narrow stick and suddenly half the test tube shattered with a bang. The sensitivity seemingly improved over time.
The pinch measuring spoon is from a 3-piece set which included a smidgen and dash. The pinch held 0.4 of water when filled with a 1 ml syringe. That
was the amount of iodine that shattered the test tube. I've another set and the 1/2 teaspoon is also engraved 2.5 ml. I used a half teaspoon of iodine
in the steel jar experiments and that was a little too perky for my neighbors probably. The pink silicone rubber band I bought at an Amazon Where Ya
Bin Store, a bag of 200 for 5 dollars!
I should say I initially started with a teaspoon of iodine and then progressed to a half teaspoon experiment and finally the pinch as my ears like
that better.
  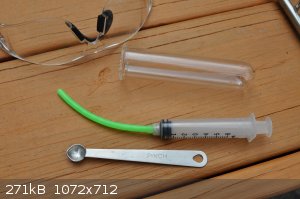
[Edited on 21-11-2025 by Morgan]
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
KNO₂ is easily made from potassium nitrate, zinc power and ammonia.
|
|
|
Radiums Lab
Hazard to Others
  
Posts: 299
Registered: 18-3-2025
Location: India
Member Is Offline
Mood: Experiencing the elegance of science.
|
|
@Keras is it pure though?
Water is dangerous if you don't know how to handle it, elemental fluorine (F₂) on the other hand is pretty tame if you know what you are doing.
|
|
|
bnull
National Hazard
   
Posts: 993
Registered: 15-1-2024
Location: East Woods
Member Is Offline
Mood: Fecking annoyed
|
|
It is safer, at least.
Edit: And cheaper. Not to mention that the DEA has its beady eyes trained on iodine.
[Edited on 22-11-2025 by bnull]
|
|
|
Radiums Lab
Hazard to Others
  
Posts: 299
Registered: 18-3-2025
Location: India
Member Is Offline
Mood: Experiencing the elegance of science.
|
|
Yea, I got it. If the purity is not of concern we can follow the method which keras pointed out.
Water is dangerous if you don't know how to handle it, elemental fluorine (F₂) on the other hand is pretty tame if you know what you are doing.
|
|
|