vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
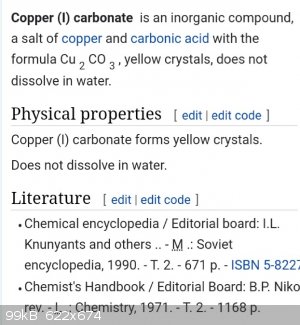
Copper (I) carbonate
Hi, today i made Copper (I) carbonate. It is a yellow solid. I thought it might have been oxide, but when I add acetic acid carbon dioxide was
released. It os a very nice and stable monovalent compound.
|
|
|
DraconicAcid
International Hazard
    
Posts: 4308
Registered: 1-2-2013
Location: The tiniest college campus ever....
Member Is Offline
Mood: Semi-victorious.
|
|
How did you make it?
Please remember: "Filtrate" is not a verb.
Write up your lab reports the way your instructor wants them, not the way your ex-instructor wants them.
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
Easily. From copper monochloride and sodium carbonate.
[Edited on 1-1-2021 by vano.kavt]
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
Now it has dark colour.

|
|
|
ChemTalk
Hazard to Self
 
Posts: 65
Registered: 13-12-2018
Location: United States
Member Is Offline
Mood: colloidal
|
|
Thanks Vano, we love how you are always coming up with very interesting compounds.
Did you mix the solids, or was sodium carbonate in solution? And did you have to heat them?
ChemTalk
chemistrytalk.org
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
Thanks. I just mixed the solutions, and soon the copper carbonate precipitated. I did not heat the solution, although I remember using warm water, but
not hot.
[Edited on 1-2-2021 by vano]
|
|
|
Maurice VD 37
Hazard to Self
 
Posts: 66
Registered: 31-12-2018
Member Is Offline
|
|
Seems to be impossible. Copper(I) chloride is not soluble into water !!! How can you mix its solution with carbonate ?
|
|
|
Bedlasky
International Hazard
    
Posts: 1227
Registered: 15-4-2019
Location: Period 5, group 6
Member Is Offline
Mood: Volatile
|
|
Copprous chloride is unstable in alkaline solution - it turn in to oxide/carbonate (depending on what makes your solution alkaline). Btw. CuCl is
soluble in slightly acidic sodium chloride solution, so this is option how to dissolve it.
[Edited on 2-2-2021 by Bedlasky]
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
In the second photo when I added acid the carbon dioxide was generated intensely. Also as far as I know chloride is soluble in both acid and alkaline
solution. However, as Bedlasky said, it is not so stable. If you are interested i can re-synthesizing in more detail.
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
[Edited on 3-2-2021 by vano]
|
|
|
Bedlasky
International Hazard
    
Posts: 1227
Registered: 15-4-2019
Location: Period 5, group 6
Member Is Offline
Mood: Volatile
|
|
Vano: If you want to dissolve CuCl, you need complexing agent, adding acid/base isn't enough.
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
In my opinion, bivalent chloride helps to dissolve, otherwise why would this carbonate precipitated.
|
|
|
Bedlasky
International Hazard
    
Posts: 1227
Registered: 15-4-2019
Location: Period 5, group 6
Member Is Offline
Mood: Volatile
|
|
CuCl dissolves in acidic, neutral or slightly alkaline solutions in the presence of complexing agents (like chlorides, bromides, sulfites,
thiosulfates, thiourea, cyanides, ammonia etc.). Copper carbonate precipitate because it have lower solubility than chloride.
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
Yes i agree with you. My cloride has greenesh colour, because it is old. I'm sure bivalent cloride helps to dissolve. It actually has a darker
color(chloride in jar).I have other chloride but it has almost the same color.
https://commons.wikimedia.org/wiki/File:Copper(I)_chloride.%E1%83%95%E1%83%99.jpg

[Edited on 4-2-2021 by vano]
|
|
|
Bedlasky
International Hazard
    
Posts: 1227
Registered: 15-4-2019
Location: Period 5, group 6
Member Is Offline
Mood: Volatile
|
|
CuCl dissolves in aqueous solutions of any soluble chlorides, not just divalent chlorides. Cation doesn't affect it.
Old samples of CuCl are always green due to aerial oxidation of CuCl in to Cu(II) oxychlorides.
|
|
|
DraconicAcid
International Hazard
    
Posts: 4308
Registered: 1-2-2013
Location: The tiniest college campus ever....
Member Is Offline
Mood: Semi-victorious.
|
|
Quote: Originally posted by Bedlasky  | CuCl dissolves in aqueous solutions of any soluble chlorides, not just divalent chlorides. Cation doesn't affect it.
Old samples of CuCl are always green due to aerial oxidation of CuCl in to Cu(II) oxychlorides. |
I suspect vano is referring to CuCl2 rather than any divalent chloride, and that will form the weird black mixed-valence complexes that so often show
up.
Please remember: "Filtrate" is not a verb.
Write up your lab reports the way your instructor wants them, not the way your ex-instructor wants them.
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
I know why it has a green color I do not see for the first time. It contains enough CuCl2 to dissolve CuCl.
[Edited on 4-2-2021 by vano]
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
which compound do you mean?
|
|
|
DraconicAcid
International Hazard
    
Posts: 4308
Registered: 1-2-2013
Location: The tiniest college campus ever....
Member Is Offline
Mood: Semi-victorious.
|
|
CuCl is insoluble in water. CuCl2 dissolves in water to give green or blue solutions, depending on concentration. A mixture of the two will give a
dark solution with a mixed-valence complex of uncertain composition.
Please remember: "Filtrate" is not a verb.
Write up your lab reports the way your instructor wants them, not the way your ex-instructor wants them.
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
Quote: Originally posted by DraconicAcid  |
CuCl is insoluble in water. CuCl2 dissolves in water to give green or blue solutions, depending on concentration. A mixture of the two will give a
dark solution with a mixed-valence complex of uncertain composition. |
Dark? Do you mean black and such colours? Or dark green....
|
|
|
woelen
Super Administrator
        
Posts: 8003
Registered: 20-8-2005
Location: Netherlands
Member Is Offline
Mood: interested
|
|
Dark in this situation is really dark. Nearly black, on very strong dilution brown/green. See
https://woelen.homescience.net/science/chem/riddles/copperI+...
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
Thanks. But it need clorine gas.
|
|
|
woelen
Super Administrator
        
Posts: 8003
Registered: 20-8-2005
Location: Netherlands
Member Is Offline
Mood: interested
|
|
No, it does not need chlorine gas. Copper(I) compounds are easily oxidized by oxygen from air, and such oxidized compounds usually have a dark
greenish or brownish color.
|
|
|
vano
National Hazard
   
Posts: 661
Registered: 22-3-2019
Location: Georgia
Member Is Offline
|
|
Okay
|
|
|