mayko
International Hazard
    
Posts: 1218
Registered: 17-1-2013
Location: Carrboro, NC
Member Is Offline
Mood: anomalous (Euclid class)
|
|
Solar-powered printing
An under-explored idea, in my opinion, is the use of solar power for direct manufacturing rather than conversion to heat or electricity.
Towards that end, I had an idea a few days ago that worked surprisingly well. My roommate and I have been building a projector, which has been a fun
project ... we took apart an computer monitor and were shining a halogen light through the LCD and focusing with a lens and mirror, up until the
screen shorted out ^_^
Anyway, we had a lot of transparency plastic lying around and a fresnel lens. So I drew on the plastic with a black sharpie and focused the light of
the sun on the plastic sheet. The clear plastic transmitted the intense light just fine, but the ink absorbs it and quickly melts the plastic! I did
some crude cutouts and made a few stencils this way.
I wonder if this could be made precise and automated, to build a solar-powered manufacturing unit?
Sorry no pix 
al-khemie is not a terrorist organization
"Chemicals, chemicals... I need chemicals!" - George Hayduke
"Wubbalubba dub-dub!" - Rick Sanchez
|
|
|
Polverone
Now celebrating 21 years of madness
        
Posts: 3186
Registered: 19-5-2002
Location: The Sunny Pacific Northwest
Member Is Offline
Mood: Waiting for spring
|
|
It has been done to fuse sand with sunlight: http://www.markuskayser.com/
PGP Key and corresponding e-mail address
|
|
|
mayko
International Hazard
    
Posts: 1218
Registered: 17-1-2013
Location: Carrboro, NC
Member Is Offline
Mood: anomalous (Euclid class)
|
|
Renewable-Energy Death Ray
I found an old DirecTV satellite dish in the overgrowth around my house. I don't have time to watch TV when there's mad science to do! So I got a
bunch of 1"x1" mirror squares and glued them to the surface of the dish, so that they focus near where the receiver was. When I take it outside, it
directs the sunlight into a focal point maybe a little bigger than a coin. On a bright day it will ignite rolled-up newspaper in seconds, and burn a
hole in bamboo within a minute or two.
Soon, I want to use it to boil a measured volume of water from a known starting temperature, to measure the wattage and the power flux.
  
al-khemie is not a terrorist organization
"Chemicals, chemicals... I need chemicals!" - George Hayduke
"Wubbalubba dub-dub!" - Rick Sanchez
|
|
|
PHILOU Zrealone
International Hazard
    
Posts: 2893
Registered: 20-5-2002
Location: Brussel
Member Is Offline
Mood: Bis-diazo-dinitro-hydroquinonic
|
|
Quote: Originally posted by mayko  | I found an old DirecTV satellite dish in the overgrowth around my house. I don't have time to watch TV when there's mad science to do! So I got a
bunch of 1"x1" mirror squares and glued them to the surface of the dish, so that they focus near where the receiver was. When I take it outside, it
directs the sunlight into a focal point maybe a little bigger than a coin. On a bright day it will ignite rolled-up newspaper in seconds, and burn a
hole in bamboo within a minute or two.
Soon, I want to use it to boil a measured volume of water from a known starting temperature, to measure the wattage and the power flux.
|
Nice idea.
Allow the beam to focus on a black stuff (charcoal) into the water otherwise much of the light (and thus potential heat) will pass through the glass
and water at the focal point at wich stade it will diverge...
PH Z (PHILOU Zrealone)
"Physic is all what never works; Chemistry is all what stinks and explodes!"-"Life that deadly disease, sexually transmitted."(W.Allen)
|
|
|
mayko
International Hazard
    
Posts: 1218
Registered: 17-1-2013
Location: Carrboro, NC
Member Is Offline
Mood: anomalous (Euclid class)
|
|
Quote: Originally posted by PHILOU Zrealone  | | Allow the beam to focus on a black stuff (charcoal) into the water otherwise much of the light (and thus potential heat) will pass through the glass
and water at the focal point at wich stade it will diverge... |
This is an important point: most of the energy is in the visible spectrum, so unreflective, dark-colored objects will absorb it more effectively.
Giving the bamboo a quick blast from a blowtorch to char the surface speeds things up quickly. I couldn't get white sugar to melt at all until I added
a grain of activated charcoal as a catalyst. Once the dark caramel products appear, things really take off.
I'm not going to burn any ants with it, but it occurs to me that it probably could power a destructive distillation of them, yielding formic acid...
Edit: The one construction flaw I've run into is: the hot glue tends to soften in the hot sun, making the mirrors liable to fall off!
[Edited on 17-7-2016 by mayko]
al-khemie is not a terrorist organization
"Chemicals, chemicals... I need chemicals!" - George Hayduke
"Wubbalubba dub-dub!" - Rick Sanchez
|
|
|
PHILOU Zrealone
International Hazard
    
Posts: 2893
Registered: 20-5-2002
Location: Brussel
Member Is Offline
Mood: Bis-diazo-dinitro-hydroquinonic
|
|
Quote: Originally posted by mayko  | Quote: Originally posted by PHILOU Zrealone  | | Allow the beam to focus on a black stuff (charcoal) into the water otherwise much of the light (and thus potential heat) will pass through the glass
and water at the focal point at wich stade it will diverge... |
This is an important point: most of the energy is in the visible spectrum, so unreflective, dark-colored objects will absorb it more effectively.
Giving the bamboo a quick blast from a blowtorch to char the surface speeds things up quickly. I couldn't get white sugar to melt at all until I added
a grain of activated charcoal as a catalyst. Once the dark caramel products appear, things really take off.
I'm not going to burn any ants with it, but it occurs to me that it probably could power a destructive distillation of them, yielding formic acid...
Edit: The one construction flaw I've run into is: the hot glue tends to soften in the hot sun, making the mirrors liable to fall off!
[Edited on 17-7-2016 by mayko] |
Use Silicon with a high temperature tolerance (up to 350°C)...that should hold even the warmest sunny days     
PH Z (PHILOU Zrealone)
"Physic is all what never works; Chemistry is all what stinks and explodes!"-"Life that deadly disease, sexually transmitted."(W.Allen)
|
|
|
crazyboy
Hazard to Others
  
Posts: 436
Registered: 31-1-2008
Member Is Offline
Mood: Marginally insane
|
|
I'm confused. Direct manufacture of what? Are you talking about using the sun to print images? SLA printers use micro-mirrors identical to those in a
DLP projector to reflect UV light in a pattern onto a photoresin. I don't know how much energy they can take before they fry but you may be able to
shine concentrated sunlight on a DLP element and reflect it onto a piece of paper for long enough to burn an image.
|
|
|
mayko
International Hazard
    
Posts: 1218
Registered: 17-1-2013
Location: Carrboro, NC
Member Is Offline
Mood: anomalous (Euclid class)
|
|
I don't have any specific target in mind; the idea I was kicking around is exploring the use of energy sources as they exist in nature, where they
exist, rather than converting them to a general-purpose form of energy like electricity and distributing them nonlocally.
For example, adding solar to the electricity grid involves some considerable inefficiency in conversion and transport:
Sunlight --(conversion loss)--> AC electricity --(transport loss)--> AC electricity --(conversion loss) --> kinetic energy, thermal energy,
etc
That's reasonable, if you live in a place or time without a lot of sunlight, or if your end goal can't be accomplished by raw sunlight. You can't cool
your house down by shining sunlight on it. But there is a certain inefficient irony in running a clothes dryer off distant sunshine, when I could just
hang my laundry out on the clothesline. The idea I'm exploring is identifying situations where it might be worth giving up the 'liquidation' and
transport of raw energy in order to reclaim the losses involved and using the raw energy in the places and times it's plentiful, for applications
where it's appropriate.
(It's not solar, but I've also wondered if local communities with wind or running water available to them might offer kinetic energy as a utility
itself - plug your coffee grinder into a pneumatic outlet instead of an electrical one! There will, of course, be some engineering kinks to work out:
https://www.youtube.com/watch?v=EXo3e58rZ7U )
Interesting idea about repurposing stereolithography printers; I'll have to check that out!
al-khemie is not a terrorist organization
"Chemicals, chemicals... I need chemicals!" - George Hayduke
"Wubbalubba dub-dub!" - Rick Sanchez
|
|
|
mayko
International Hazard
    
Posts: 1218
Registered: 17-1-2013
Location: Carrboro, NC
Member Is Offline
Mood: anomalous (Euclid class)
|
|
I measured a time series of temperatures of 100 mL water with a spoonful of charcoal, held approximately in the focus:
| Code: |
minutes seconds time temp
0 0 0 17
1 20 80 31
2 30 150 37
3 30 210 44
4 40 280 52
6 0 360 58
7 20 440 57
8 30 510 56
9 10 550 57
10 50 650 61
13 0 780 74
14 30 870 81
16 40 1000 91
19 20 1160 96
|
Graphing these data, we can see where a cloud blew through and obscured the sun:
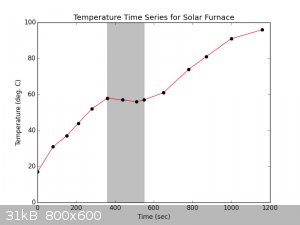
We can fit a linear regression to the whole time series to get an idea of the approximate power delivery on a decently bright day. We can also fit
regressions to the two sunny periods to get an idea of the power delivery at peak sunlight.
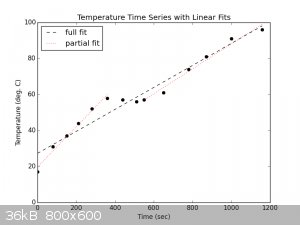
Using the fit parameters and a heat capacity of 4.1796 J/(K*cm**3), the wattage delivered can be calculated:
| Code: |
m (deg C/sec) b (deg C) power delivered (Watts)
full 0.061073826 27.2885906 25.52641611
sunny1 0.111893764 19.69245574 46.76711778
sunny2 0.069095229 18.97215073 28.87904177
|
The solar collector works by concentrating sunlight; we can thus use the number of 1in*1in mirrors in the reflectors (230 count), the measured size of
the flask bottom (diameter 8cm), and the approximate size of the focus (1inx1in) to calculate the incoming power flux, the measured power flux, and
the power flux at the focus, respectively:
| Code: |
flux at dish (W/m**2) flux at flask (W/m**2) flux at focus (W/m**2)
full 172.0261917 5074.834216 39566.0241
sunny1 315.1703371 9297.63773 72489.17754
sunny2 194.6200186 5741.360194 44762.60427
|
Wikipedia relays this from the WMO:
| Quote: |
The World Meteorological Organization uses the term "sunshine duration" to mean the cumulative time during which an area receives direct irradiance
from the Sun of at least 120 watts per square meter.
|
So the figures incoming power flux at the dish are at least in the ballpark.
[Edited on 1-8-2016 by mayko]
al-khemie is not a terrorist organization
"Chemicals, chemicals... I need chemicals!" - George Hayduke
"Wubbalubba dub-dub!" - Rick Sanchez
|
|
|
PHILOU Zrealone
International Hazard
    
Posts: 2893
Registered: 20-5-2002
Location: Brussel
Member Is Offline
Mood: Bis-diazo-dinitro-hydroquinonic
|
|
Very nice work Mayko!
There is a slight decay into the slope close to 100°C because some water is already vapourizing even if not boiling yet  . .
Now you can turn this into dynamic energy/electricity with help of a Stirling motor  
[Edited on 1-8-2016 by PHILOU Zrealone]
PH Z (PHILOU Zrealone)
"Physic is all what never works; Chemistry is all what stinks and explodes!"-"Life that deadly disease, sexually transmitted."(W.Allen)
|
|
|
Pyro_cat
Hazard to Others
  
Posts: 243
Registered: 30-4-2018
Member Is Offline
Mood: No Mood
|
|
I wrote to a nuclear physicist once after reading and article on fusion asking why they don't try concentrating sunlight to create enough heat to get
the reaction going. He wrote back and told me it would take a magnifying glass 300 Kilometers across.
|
|
|
RogueRose
International Hazard
    
Posts: 1585
Registered: 16-6-2014
Member Is Offline
|
|
Try to find something dark green, like chlorophyll, from all the stuff I've read, mixing a green with the black (which you can't really see) increases
the amount of absorbed light & heat.
|
|
|
Metacelsus
International Hazard
    
Posts: 2531
Registered: 26-12-2012
Location: Boston, MA
Member Is Offline
Mood: Double, double, toil and trouble
|
|
Quote: Originally posted by Pyro_cat  | I wrote to a nuclear physicist once after reading and article on fusion asking why they don't try concentrating sunlight to create enough heat to get
the reaction going. He wrote back and told me it would take a magnifying glass 300 Kilometers across.
|
Your physicist friend should have realized a purely passive optical setup such as that one could only heat something to the temperature of the surface
of the sun, which is far too low for fusion.
|
|
|
Pyro_cat
Hazard to Others
  
Posts: 243
Registered: 30-4-2018
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by Metacelsus  | Quote: Originally posted by Pyro_cat  | I wrote to a nuclear physicist once after reading and article on fusion asking why they don't try concentrating sunlight to create enough heat to get
the reaction going. He wrote back and told me it would take a magnifying glass 300 Kilometers across.
|
Your physicist friend should have realized a purely passive optical setup such as that one could only heat something to the temperature of the surface
of the sun, which is far too low for fusion. |
I wrote to the guy that wrote the article but I did think of it when posting that. I read about that limitation forgot what it was called. cause the
sun is round right ?
Maybe fusion needs a critical mass so just pin pointing less then 300km round area at a few atoms wouldn't do it.
|
|
|
VSEPR_VOID
National Hazard
   
Posts: 719
Registered: 1-9-2017
Member Is Offline
Mood: Fullerenes
|
|
Glass making is a very complicated process. There have been thousand of years of scientific history surrounding it. I recall that in some European
countries a few hundred years ago, the making of specific glasses (known for their optical quality), was a state secret; kept under armed guard. This
ended up costing Micheal Faraday several years of his life when he was assigned the task of reverse engineering some variety of foreign glass.
Applied Science, peace be upon him, has a video on it. Of course he would, the glorious bastard also made a scanning electron microscope from bubble
gum and tooth picks:
https://www.youtube.com/watch?v=mUcUy7SqdS0
I would think that just hooking up a giant Fresnel lens to a giant CNC machine and sticking it in the desert to make gaint glass structures, well
being fun, would yield inconsistent results. The glass structures it would sinter would be very unreliable in quality due to inconsistencies in the
sand.
Within cells interlinked
Within cells interlinked
Within cells interlinked
|
|
|