Alchemica
Harmless

Posts: 25
Registered: 31-5-2025
Member Is Online
Mood: No Mood
|
|
Crossed-aldol formation of dehydrozingerone and an experimental attempt at a novel hydrogenation
My chemistry is VERY rusty so forgive me but if anyone has tried anything along similar lines for hydrogenation, I'd love to hear.
In a quest to create some new ginger components, starting with dehydrozingerone and it's derivatives like zingerone (pungent, spicy aromatic) and
zingerol (non-pungent), I've been looking for novel, simple and accessible ways to explore catalytic hydrogenation. Where I am, platinised titanium
mesh is easily available and I wanted to see if it could have suitable catalytic activity.
The crossed-aldol reaction on aromatic aldehydes proceeds as here
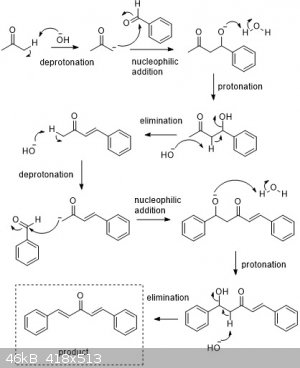
Dehydrozingerone
Curcumin is limited in translatable clinical effectiveness in part due to it's poor pharmacokinetic properties. Dehydrozingerone is a curcumin
degradant which is a half structural analog of curcumin. It exists as a natural phenolic compound obtained from rhizomes of Zingiber officinale, which
has attracted much attention of medicinal chemists. It is known to have a broad range of biological activities like antioxidant, anticancer,
anti-inflammatory, anti-depressant, anti-malarial, antifungal, anti-platelet and many others.
Crossed-aldol reaction of vanillin and acetone
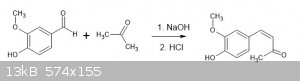
The following procedure [1] was scaled up:
In a 50 mL round-bottom flask, add 0.5 g (3.3 mmol) of vanillin then 5 mL (67.5 mmol) of acetone with vigorous stirring. After vanillin has dissolved
completely (usually very fast) add 2.63 mL NaOH 2.5 M (the concentration of NaOH is important as using more concentrated NaOH will produce a thick
paste mixture which extends reaction time and further complicates the precipitation process).
Allow to stand overnight and then dropwise add dilute acid (2.5 mL 6M HCl etc). Cool in a freezer and collect the yellow shards of dehydrogzingerone.
Attempted novel reduction
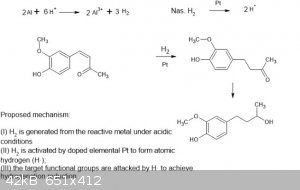
As a novel reduction experiment, a small-scale novel experiment to attempt to reduce the α,β-unsaturated carbonyl compound was conducted using
dehydrozingerone. I can't find much on similar techniques in the literature, other than doing similar with Raney Nickel coupled with electrochemically
generated nascent H2 [2]. Wasn't sure if the platinised titanium would have sufficient surface area vs something like Pd/C etc to work, but it looks
like it converts the dehydrozingerone to new Rf constituents.
The materials for the experiment

Reaction vessel set-up

It seems to (?) be quite effective on the 0.5g scale with the compound dissolved in 50% aqueous ethanol. TLC indicates complete reduction of
conjugated fluorescent compounds (? divanillylideneacetone impurity in starting product?) and formation of new, similar but lower Rf potentially
unsaturated constituents
TLC - sorry for poor image

Left lane (A): dehydrozingerone (B) Reaction mixture after novel hydrogenation attempt. Viewed under UV 395nm – first plate and I2 visualisation
– second plate.
Olfactorily, there is a formation of a new ginger scent and loss of the yellow colouration of the starting material
It's as simple as some folded platinised titanium mesh as the catalyst, allowing for easy and complete catalyst recovery, whilst generating nascent
hydrogen in situ from dissolving aluminium strips and gradual addition of HCl, near the mesh. It's very friendly and simple to do
[1] “Crossed Aldol Reactions in Water Using Inexpensive and Easily Available Materials as a Tool for Reaction Optimization Teaching in an
Undergraduate Organic Chemistry Laboratory” Kevin A. Ruiz, Marta López, Gottfried Suppan, and Kamil Makowski Journal of Chemical Education 2023 100
(10), 4160-4160 DOI: 10.1021/acs.jchemed.3c00902
[2] Wang W, Niu J, Yang Z. An efficient reduction of unsaturated bonds and halogen-containing groups by nascent hydrogen over Raney Ni catalyst. J
Hazard Mater. 2020 May 5;389:121912. DOI: 10.1016/j.jhazmat.2019.121912.
[Edited on 31-5-2025 by Alchemica]
|
|
|
davidfetter
Hazard to Self
 
Posts: 60
Registered: 8-9-2021
Member Is Offline
|
|
Any chance you could replace those references to an external site with attachments? We've lost a lot over the years to sites that seemed like they'd
be up much longer than they actually were.
|
|
|
Alchemica
Harmless

Posts: 25
Registered: 31-5-2025
Member Is Online
Mood: No Mood
|
|
Quote: Originally posted by davidfetter  | | Any chance you could replace those references to an external site with attachments? We've lost a lot over the years to sites that seemed like they'd
be up much longer than they actually were. |
Sorry, taking me a bit to work out how to properly add pics here, hopefully that's better now. Thanks for the tip
|
|
|
davidfetter
Hazard to Self
 
Posts: 60
Registered: 8-9-2021
Member Is Offline
|
|
Quote: Originally posted by Alchemica  | Quote: Originally posted by davidfetter  | | Any chance you could replace those references to an external site with attachments? We've lost a lot over the years to sites that seemed like they'd
be up much longer than they actually were. |
Sorry, taking me a bit to work out how to properly add pics here, hopefully that's better now. Thanks for the tip |
Thanks for helping future readers see the cool stuff you're doing!
|
|
|
Alchemica
Harmless

Posts: 25
Registered: 31-5-2025
Member Is Online
Mood: No Mood
|
|
While I have some Pd/C these days, for the sake of amateur chemistry:
Found these REALLY cheap Pt plated beads ($4.95 AU per 500, 2mm Pt plated glass beads). They mag stir really well so curious if they could add another
dimension added to dissolving metal reductions eg Zn/HCl eg if the nascent H2 would absorb onto the Pt surface for an added catalytic
hydrogenation effect.
Work in progress.

|
|
|