Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
Two failed experiments: 4-aminophenol and benzocaine
Folks,
I tried two distincts experiments this weekend, and they both failed, and I was curious to know if you had thoughts to share about any of them.
Benzocaine: this is probably the simplest of all. Basically a Fisher ester synthesis between PABA and ethanol. But I can’t get it to work. Even with
a large excess of ethanol (15 g PABA, 50 mL 99% ethanol, 1.5 mL 70% methanesulphonic acid) and two hours cooking, it doesn’t work. With that
combination, when the mixture is poured into 250 mL of sodium carbonate saturated water, the crystals (very nice btw) I get after cooling turn out in
the end only to be hydrated PABA (they melt around 40 °C releasing their water of crystallisation). True, there is a sort of plateau around 90 °C,
but nothing really melts, and after boiling off the water I’m left with a solid white powder which: 1. doesn’t melt around 90 °C as expected; and
2. has no perceptible numbing properties. It must be unchanged PABA.
Now I’m wondering whether all the acid doesn’t go into the protonation of the PABA NH₂ group, leaving effectively no free protons, which would
explain the failure. Do I have to add a little over the stoichiometric amount of strong acid so as to protonate all the amine group of the PABA? In
hindsight, that would make sense.
4-aminophenol: This is normally made by reducing 4-nitrosophenol (obtained by nitrosation of phenol under 0 °C, this step works fine) with sodium
hydrosulphite. Problem: albeit that all the brown/orange colour characteristic of 4-nitrosophenol disappears as expected, and is replaced by a pale
yellow somewhat turbid solution, I can’t get anything to crystallise out of it, even at pH=7, at which the solubility of 4-aminophenol is said to be
minimal.
After a couple of hours in the fridge, there is a slight powdery layer at the bottom of the beaker, but it’s only a handful of milligrams (out of 10
g phenol), that turns from white to bluish when exposed to the air. I saturated the solution with salt, to no avail. Finally I tried to extract the
4-aminophenol from water using DCM, and got something somewhat unexpected: while the DCM layer is colourless when it collects at the bottom of the sep
funnel, once exposed to air it immediately turns into an intense fuchsia/purple coloured liquid reminiscent of either iodine or potassium permanganate
(see picture). Boiling the DCM off, I recovered a handful of small dark crystals, which do not really dissolve in water but colour it yellow. When
acid is added, the yellow turns to red. When the solution is made basic, it turns to blue, then somewhat brown. What kind of pH indicator can this be?
Maybe 4-aminophenol is not soluble in DCM? I should’ve tried diethyl ether, or ethyl acetate instead.

[Edited on 20-10-2025 by Keras]
|
|
|
Maui3
Hazard to Others
  
Posts: 173
Registered: 9-9-2024
Member Is Offline
|
|
I have tried the same experiment a few years ago with sodium bisulfate instead of methanesulfonic acid or sulfuric acid. I really just think that you
have to use sulfuric acid for it.
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
Quote: Originally posted by Maui3  | | I have tried the same experiment a few years ago with sodium bisulfate instead of methanesulfonic acid or sulfuric acid. I really just think that you
have to use sulfuric acid for it. |
Oh, my second try was made with sulphuric acid (98%). It failed in the same manner.
|
|
|
Fery
International Hazard
    
Posts: 1117
Registered: 27-8-2019
Location: Czechoslovakia
Member Is Offline
|
|
How much H2SO4 did you use? The amino group of the PABA could bind the H2SO4 so not enough acidity for the catalytical reaction.
[Edited on 20-10-2025 by Fery]
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
Quote: Originally posted by Fery  | How much H2SO4 did you use? The amino group of the PABA could bind the H2SO4 so not enough acidity for the catalytical reaction.
[Edited on 20-10-2025 by Fery] |
Yeah, that’s what I reasoned in hindsight. In each experiment, I used only a couple of mL of acid (catalytic amount), which might be insufficient.
Next time I’ll check the pH and ensure it is ~ 1 before refluxing.
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
So I retried the synthesis using what I thought was PABA, but it turns out it was mainly sodium carbonate I had used to basify the fresh water I had
poured the mixture into.
Anyways, there must've been a few milligrams of leftover PABA in it. This time I used an excess of acid, household alcohol, and got, after 90 min
refluxing, pouring into cold water, raising the pH to 10 using sodium hydroxide and leaving the erlenmeyer in the fridge overnight, the result you see
in the picture. While the big crystals are, I think, hydrated sodium sulfate, can the few yellowish/orange flakes seen at the bottom be benzocaine?

|
|
|
bnull
National Hazard
   
Posts: 993
Registered: 15-1-2024
Location: East Woods
Member Is Offline
Mood: Fecking annoyed
|
|
Not 4-nitrophenol? Did you follow any procedure from, say, Org. Syn.? I have to admit it is the first time I hear of 4-aminophenol by reduction of
something other than 4-nitrophenol.
The way the "indicator" works suggests me a quinone. See if reacts with KI+HCl.
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
Quote: Originally posted by bnull  |
Not 4-nitrophenol? Did you follow any procedure from, say, Org. Syn.? I have to admit it is the first time I hear of 4-aminophenol by reduction of
something other than 4-nitrophenol.
The way the "indicator" works suggests me a quinone. See if reacts with KI+HCl. |
Interesting.
Nitrosation is taken from this pathway (see picture).

|
|
|
bnull
National Hazard
   
Posts: 993
Registered: 15-1-2024
Location: East Woods
Member Is Offline
Mood: Fecking annoyed
|
|
| Quote: | | Nitrosation is taken from this pathway (see picture). |
Where's it from?
Meanwhile, does your nitrosophenol look like this (Merck Index 14th)?

|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
I thought I had the source PDF of this paper document but I can’t seem to find it. 
Turning back to p-nitrosophenol, well, not really. What I do get is a bright orange/brownish powder in a yellow liquid that I filter off. I never
bothered recrystallising it. (I drip sodium nitrite into an acidic solution of phenol.)
I’m not particularly suspicious about nitrosation. It is fairly well documented, and one of the main advantages of nitrosation over nitration is the
production of almost pure p-nitrosophenol, whereas nitration privileges the ortho isomer. Vogel mentions the reaction though, although it later
mentions also that p-nitrosophenol can bind to unreacted phenol to form indophenol (see attachments).
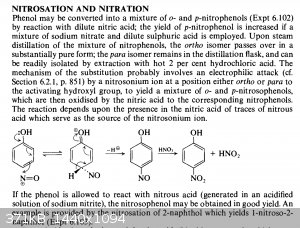 
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
So I redid the benzocaine experiment with fresh PABA I ordered (pills), household cleaning alcohol and enough 37% sulphuric acid to get the mixture
really acidic (pH < 1) and I think it somewhat worked. Of course, I didn't get such a high yield given how much water was present from the get go,
but at least I've got a product that seems to have a numbing effect. I’m going to assess the melting point later this morning.
The capture of protons by the NH₂ group is something to take into account. In hindsight, it explains why my previous attempt at making methyl
anthranilate failed miserably.
[EDIT: Picture of benzocaine after recrystallisation from boiling water.]
[Edited on 23-10-2025 by Keras]

|
|
|
Dr.Bob
International Hazard
    
Posts: 2917
Registered: 26-1-2011
Location: USA - NC
Member Is Offline
Mood: Mildly disgruntled scientist
|
|
That is great. This is one case where full protonation of the amino group is needed before much else will happen, I would likely try to use 2 eq of
acid. The amount of water and other bits (like denaturants) will lower the yield, but sounds like it worked enough. And trying to do the coupling
with other methods would likely cause side effects from the amino group making the amide polymer.
Lidocaine is not too hard to make also, but requires chloroacetylchloride for the easiest route, and that stuff is hard to make and keep stable in
storage.
|
|
|
Lionel Spanner
Hazard to Others
  
Posts: 203
Registered: 14-12-2021
Location: near Barnsley, UK
Member Is Offline
|
|
In a previous job I occasionally used to work with 4-aminophenol, as component of oxidative hair dyes. It's water-soluble, so I can't imagine it'd be
very soluble in DCM.
[Edited on 24-10-2025 by Lionel Spanner]
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
Yeah, the solubility is 11 g/L even at 0 °C. Since I usually start with 10 mmol of phenol, and M = 109.3 g/mol for 4-aminophenol, that means 100 mL
is enough to dissolve all of it (and I suppose the figure is valid for pH = 7 which is the pH at which the solubility is minimal).
Even when salted, the solution does not produce any noticeable amount of product. I wonder how I can extract it then. Ethyl acetate?
|
|
|
Lionel Spanner
Hazard to Others
  
Posts: 203
Registered: 14-12-2021
Location: near Barnsley, UK
Member Is Offline
|
|
Quote: Originally posted by Keras  | Yeah, the solubility is 11 g/L even at 0 °C. Since I usually start with 10 mmol of phenol, and M = 109.3 g/mol for 4-aminophenol, that means 100 mL
is enough to dissolve all of it (and I suppose the figure is valid for pH = 7 which is the pH at which the solubility is minimal).
Even when salted, the solution does not produce any noticeable amount of product. I wonder how I can extract it then. Ethyl acetate?
|
Methyl isobutyl ketone might be worth a try - it's chemically similar to acetone but immiscible with water. It's good for difficult compounds that
aren't soluble in less polar solvents.
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
Oh, that’s a good idea. I think I have a bottle of methyl-tert-butyl ether, maybe it could do the trick? Seems to be even less soluble than methyl
isobutyl ketone.
I also have diethylene glycol butyl ether, but I think that one is water soluble. So is THF. 
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
So, follow-up. I redid the 4-aminophenol experiment following more or less the protocol written on the paper (especially the quantity of sodium
hydrosulfite). The brownish crystals dissolve after the sodium hydrosulfite addition and the solution turns to green (picture). It is stirred for 10
min at 45 °C then cooled and filtered.
When filtered, the funny thing is that the solution to filter is green, but the filtrate is yellow (see picture).
I recrystalled the precipitate and got a crop of nice colourless crystals. I was somehow unsure this was 4-aminophenol, but the crystals are browning
by the minute (see pictures), as it said Rodinal™ does, so this must be the real deal. I’ll try to transform it into acetaminophen tomorrow using
reductive acylation with acetic acid and zinc and see what's coming off it. Yet, I'd be interested if anyone had a means to test that this is really
4-aminophenol.
[EDIT] Because I fear the crystals are just some sodium salt covered in a thin layer of 4-aminophenol.
---
PS: I suppose p-nitrosophenol is first reduced to phenolhydroxylamine (OH-⏣-NO → OH-⏣-NHOH) and only then phenolhydroxylamine to 4-aminophenol
OH-⏣-NHOH → OH-⏣-NH₂ + H₂O. My mistake was probably to not use enough sodium hydrosulfite, which would’ve blocked the reduction at phase
1.
   
[Edited on 26-10-2025 by Keras]
|
|
|
Lionel Spanner
Hazard to Others
  
Posts: 203
Registered: 14-12-2021
Location: near Barnsley, UK
Member Is Offline
|
|
Try dissolving some of it in water, then add in some hydrogen peroxide and make the solution basic, either with hydroxide or a suitable amine, and
leave it to stand for a few minutes. If it's 4-aminophenol, a dark brown/black insoluble compound will slowly form.
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
Thanks!
TBH, after much mulling and grumbling, I’m going to buy a bottle of Rodinal™, the photographic developing agent. It’s basically 4-aminophenol,
so I’ll be able to run a TLC and see if this is the real McCoy!
|
|
|
chempyre235
Hazard to Others
  
Posts: 207
Registered: 21-10-2024
Location: Between Nb and Tc
Member Is Offline
Mood: Quite distracted
|
|
I found that Adorama sells the pure chemical for $50/lb or $15/100g. Interestingly, it's sourced from the Photographers' Formulary, but I can't find it on
their website. Maybe I just missed it?
"However beautiful the strategy, you should occasionally look at the results." -Winston Churchill
"I weep at the sight of flaming acetic anhydride." -@Madscientist
"...the elements shall melt with fervent heat..." -2 Peter 3:10
|
|
|
Keras
International Hazard
    
Posts: 1143
Registered: 20-8-2018
Location: (48, 2)
Member Is Offline
|
|
Sometimes businesses sell only in B to B mode, because they don’t want the extra hassle of dealing with small quantities.
|
|
|
dicyanin
Hazard to Self
 
Posts: 69
Registered: 29-3-2020
Location: Europe
Member Is Offline
Mood: inquisitive
|
|
extraction of PABA and benzocaine synthesis
EXTRACTION OF PABA FROM SUPPLEMENT PILLS
100 pieces of PABA 500 mg pills1 were ground in an electric coffee grinder, the powder was placed in a beaker and covered with 400 ml hot
denatured ethanol2. It was allowed to stand overnight with occasional stirring, filtered through 2 coffee-filters, and the filtrate
evaporated until 1/3rd of the original volume. After cooling in the refrigerator for a few hours the product precipitated. The large needles of
4-aminobenzoic acid were filtrated and air dried.
Yield: 40 g (80%)3
[1] other ingredients: microcrystalline cellulose, calcium phosphate, corn starch, magnesium stearate, silica gel
[2] denaturant : 2-butanone (5%)
[3] an additional crop of 6 g solids was obtained by evaporating the solvent to dryness but these were considered too impure so these were discarded

SYNTHESIS OF BENZOCAINE
In a 1L roundbottom flask was added 5.74 g (0.042 mol) 4-aminobenzoic acid and 75 ml denatured ethanol. The mixture was stirred until a clear solution
was obtained, after which 5 ml concentrated sulfuric acid was added dropwise. A voluminous precipitate of the PABA sulfate salt was observed.
The mixture was refluxed for 8 hours, during which the solids gradually went into solution as the reaction progressed. After cooling the clear
solution to room temperature a yellow precipitate was noticed. The flask was placed in an ice/waterbath and with good stirring there was added
carefully, in small increments 200 ml saturated aqueous sodium bicarbonate solution.
The precipitate was collected through filtration* and the filter cake washed with small amounts of water, and dried in a dessicator over anhydrous
calcium chloride for 48 hours.
Yield: 9.90 g crude benzocaine sulfate as an offwhite amorphous powder
(90%)
Taste test of a few milligrams: definite and undeniable local anaesthesia of the tongue
odor: pleasant and vanilla-like
(*) I checked the pH of the filtrate and it was ~8 to indicator paper.
I expected to obtain benzocaine as a base after basifying with bicarbonate, but judging from the amount it must be the sulfate salt.
I haven't had the change to do a recrystallisation, nor a melting point of the base, will follow up on this.

sic transit gloria mundi
|
|
|
dicyanin
Hazard to Self
 
Posts: 69
Registered: 29-3-2020
Location: Europe
Member Is Offline
Mood: inquisitive
|
|
Quote: Originally posted by Keras  |
Benzocaine: this is probably the simplest of all. Basically a Fisher ester synthesis between PABA and ethanol. But I can’t get it to work. Even with
a large excess of ethanol (15 g PABA, 50 mL 99% ethanol, 1.5 mL 70% methanesulphonic acid)
[Edited on 20-10-2025 by Keras] |
Pretty sure your mistake was not calculating in the amine salt formation, you used not enough acid.
15 g PABA = 0.11 mol
1.5 ml mesylic acid 70% = 1.319 g/ml according to Sigma-Aldrich datasheet, so 1.319 g/ml * 1.5 ml = 1.979 g
1.979 g 70% mesylic aid = 1.979 * 0.70 = 1.385 g mesylic acid
mol. wt. mesylic acid = 96.10 g/mol so 1.385 / 96.10 = 0.014 mol
so far too little acid, it just makes the amine salt, there is no excess present to even let the Fischer esterification to proceed. You have basically
just been refluxing a mixture of PABA and PABA mesylate in ethanol.
I would try again but instead use 0.15 or better 0.20 mol mesylic acid, so 19.22 g / 0.70 = 27 g 70% methanesulfonic acid for 15 g of PABA
Also do your basification more gentle, in aliquots, and with external cooling, don't just dump it in sodium carbonate solution. Treat it like a woman

sic transit gloria mundi
|
|
|