Fery
International Hazard
    
Posts: 1129
Registered: 27-8-2019
Location: Czechoslovakia
Member Is Offline
|
|
crown macromolecule synthesis - 2,2,7,7,12,12,17,17-Octamethyl-21,22,23,24-tetraoxaquaterene
In 250 ml FBF (100 ml should be enough) in ice cold water bath was mixed 0,40 g anhydrous LiCl, 8 ml 35% HCl (LiCl fully dissolved), 0,80 ml 70% HClO4
(to make 1,0 g LiClO4 in situ). Then 15 ml 95% ethanol (free of butanone) was added. Then 7 ml furan (0,1 mol) and 15 ml acetone (0,2 mol) was added
while mixing. Clear colorless homogenous reaction mixture obtained. Flask stoppered and put aside. After 10 minutes pinkish coloration, after 20
minutes more pink color. After 4 hours already some crystalline product (small amount). The mixture will be let to react undisturbed at room
temperature for 2 weeks.
LiClO4 is one of the best soluble from all Li salt in organic polar solvents. The Li cation facilitates 4 furan member ring cyclization. The reaction
runs without Li cation too, just much slower and very likely also 5 and 6 furan member molecules are produced.
Пожарский А.Ф., Анисимова В.А., Цупак Е.Б.
Практические работы по химии гетероциклов
http://www.newlibrary.ru/read/pozharskii_a_f___anisimova_v_a...
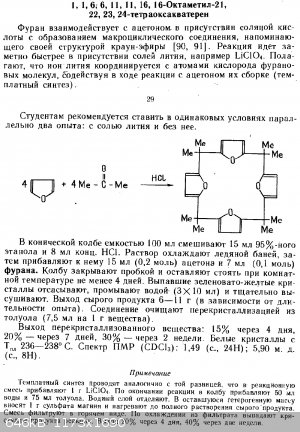
more advanced and more accurate synthesis:
https://www.orgsyn.org/demo.aspx?prep=CV6P0856
Attachment: CV6P0856.pdf (330kB)
This file has been downloaded 29 times
Attachment: kobuke1976.pdf (740kB)
This file has been downloaded 33 times
Attachment: brown-sawatzky-2011-the-condensation-of-furan-and-sylvan-with-some-carbonyl-compounds.pdf (263kB)
This file has been downloaded 17 times
after 10 minutes pinkish color

after 20 minutes pink color

after 4 hours already some product, the reaction is very slow and facilitated by Li cation which has a template effect to create 4 furan ring crown
molecule, the reaction will be sit at room temperature for 2 weeks further

[Edited on 14-3-2026 by Fery]
|
|
|
Texium
Administrator
       
Posts: 4748
Registered: 11-1-2014
Location: Salt Lake City
Member Is Offline
Mood: Seeking gainful employment
|
|
Nice! I’ve been interested in the process of templating crown ether formation using alkali cations for a long time, but this is the first time
I’ve seen someone actually do it in practice. Looking forward to seeing your final results!
|
|
|
Boffis
International Hazard
    
Posts: 1927
Registered: 1-5-2011
Member Is Offline
Mood: No Mood
|
|
Nice experiment Fery! Does this cyclic ether have complexing abilities with alkali and alkaline earth ions like the polyethylene oxide type ether? I
look forward to the results too.
|
|
|
Fery
International Hazard
    
Posts: 1129
Registered: 27-8-2019
Location: Czechoslovakia
Member Is Offline
|
|
Hello, after 1 day the mass in flask was already quite thick, yellow-green colored.
In the attached kobuke.pdf there is the Table I. with data of extraction of Li+, Na+, K+, Rb+, Cs+, NH4+, Ag+ picrates from aqueous to organic phase.
I already left my lab for 2 weeks, that's why I started the experiment just before leaving.
If using bigger cation, maybe Ag+, NH4+, Cs+, Rb+ maybe 5 or 6 membered ring will be formed? This is a template reaction so the molecule binds around
the cation, the bigger cation the bigger macromolecule. Li facilitates 4 membered ring formation. But the reaction slowly proceeds even without any
cation. If you have only 4 days time then certainly use a cation. If you have 2 weeks time then if you do not have any cation you may try it that way.
I do not know whether LiCl instead of LiClO4 will dissolve, if not fully maybe at least partially will dissolve?
Here photos 1 day after starting the reaction.
 
|
|
|
chempyre235
Hazard to Others
  
Posts: 216
Registered: 21-10-2024
Location: Between Nb and Tc
Member Is Offline
Mood: Quite distracted
|
|
Most impressive! I've been wanting to do some experiments with crown ethers as well. IIRC, (at least with the poly-ethylene oxide crowns) sodium
cations form a 5-unit ring, potassium is a 6, rubidium is a 7, et cetera. I hadn't even considered using furans, nor did I know it could be done so
easily! Excellent work!
I'm wanting to work with some thiacrown ethers, as they're supposedly more selective toward transition metals, instead of alkali metals.
@Fery, I'm curious: where did you get your furan, and how much does it tend to run? I see Fischer carries 250ml for about $70 US. Is this close to the
average cost?
[Edited on 3/16/2026 by chempyre235]
"However beautiful the strategy, you should occasionally look at the results." -Winston Churchill
"I weep at the sight of flaming acetic anhydride." -@Madscientist
"...the elements shall melt with fervent heat..." -2 Peter 3:10
|
|
|
Fery
International Hazard
    
Posts: 1129
Registered: 27-8-2019
Location: Czechoslovakia
Member Is Offline
|
|
Hi Chempyre, I've bought it in bulk with other chemicals, it was like 100 old (but well stored) chemicals of 100 kg for 1000 EUR, some of them still
unopened, or if already opened then usually almost full (only little used). My bottle of furan has 1 L. It could be like 10-20 years old but still in
good condition (I did not redistill it). I was happy that I finally found a usage for it.
Furan can be made from furfural (from corncobs and other poly pentoses, like cherry resin) by oxidation to furoic acid and then decarboxylation. But
I'm not sure whether the decarboxyllation runs also in small scale lab or whether the route is suitable only for industry. It has very low b.p. even
lower than diethylether, you need very efficient condenser and very cold circulating liquid.
|
|
|
Chem Science
Hazard to Others
  
Posts: 127
Registered: 30-7-2018
Location: Argentina
Member Is Offline
|
|
Nice work Fery !
Inspired by your work, I carried out some experiments to see if I could make similar compounds. I tried using 3-methylthiophene (3-MT) and
2,5-dimethylfuran. Unfortunately, I didn’t obtain any products. The first result makes sense—I didn’t expect any reaction due to the methyl
groups blocking the reactive positions, but I wanted to see what would happen (the solution darkened over time and formed a brown precipitate).
With 3-MT, I refluxed the reaction and added extra ethanol to improve solvation, but TLC showed new compounds forming without any precipitate.
Anyway, I thought it was interesting. If I can obtain furan, I will reproduce your work—I like crowns 
|
|
|
chempyre235
Hazard to Others
  
Posts: 216
Registered: 21-10-2024
Location: Between Nb and Tc
Member Is Offline
Mood: Quite distracted
|
|
It looks like it may be feasible to get furan from table sugar. According to this paper, vacuum pyrolysis at 500°C affords over 50% conversion to furan derivatives. Most of these compounds are 5-hydroxymethylfurfural
(HMF), furfural, and some more minor products which are not useful here. Oxidation of HMF and furfural with KMnO4 should yield acid
derivatives, which are decarboxylated relatively easily, I believe. In the lab, I'd assume that lower temps would be used with sulfuric acid for the
dehydration step.
@Chem Science, I agree with your analysis with the reactants you chose, but those methyl groups are relatively active, and should also be subject to
chemical oxidation. You may be able to get furan that way as well.
Anyway, I don't want to clog up this thread with a tangential topic any more than I already have, so if I come up with anything else, I'll add it to
the furan thread or start a new one.
"However beautiful the strategy, you should occasionally look at the results." -Winston Churchill
"I weep at the sight of flaming acetic anhydride." -@Madscientist
"...the elements shall melt with fervent heat..." -2 Peter 3:10
|
|
|
|