Gioacchino
Harmless

Posts: 4
Registered: 22-5-2025
Member Is Offline
Mood: No Mood
|
|
reaction between 2-methyl-2-cyclohexenone and an excess of cyanide ion, in the presence of hydrochloric acid
Hi everyone, I can’t understand the mechanism of this reaction. First of all I don’t get it why cyanide ion doesn’t react with ketone and why
instead hydrocianic acid reacts with the double bond. But also in this case I don’t understand why the electrophile hydrogen atom doesn’t follow
the Markovnikov’s rule. Beacause then the ciano group will be on a different carbon atom than I expect. Thanks
|
|
|
Alchemica
Harmless

Posts: 26
Registered: 31-5-2025
Member Is Offline
Mood: No Mood
|
|
I'm guessing you have predominately a 1,4-addition to the enone?
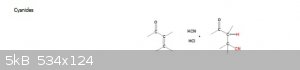
As the nucleophile, the cyanide anion, is a weak base, then the 1,2 addition is usually reversible. This means the competition between 1,2 and 1,4
addition is under thermodynamic control. In this case 1,4-addition dominates because the stable carbonyl group is retained.
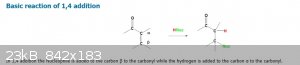
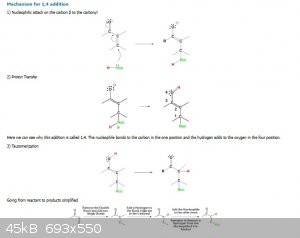
See: https://chem.libretexts.org/Bookshelves/Organic_Chemistry/Map%3A_Organic_Chemistry_(Vollhardt_and_Schore)/18%3A_Enols_Enolates_and_the_Aldol_Condensat
ion%3A_ab-Unsaturated_Aldehydes_and_Ketones/18.10%3A_12-_and__14-Additions_of_Organometallic_Reagents
|
|
|