| Pages:
1
..
33
34
35
36
37
38 |
Abromination
Hazard to Others
  
Posts: 432
Registered: 10-7-2018
Location: Alaska
Member Is Offline
Mood: 1,4 tar
|
|
Quote: Originally posted by Jackson  | Cracked my beaker while cleaning off burnt nitrates and pushing to hard while scraping. one of my Erlenmeyer flasks cracked internally from heat
shock. Like the cracking is inside the glass and doesnt actually go through the walls.
|
Even if it is only cracked internally, you no longer should use it as it could shatter.
List of materials made by ScienceMadness.org users:
https://docs.google.com/spreadsheets/d/1nmJ8uq-h4IkXPxD5svnT...
--------------------------------
Elements Collected: H, Li, B, C, N, O, Mg, Al, Si, P, S, Fe, Ni, Cu, Zn, Ag, I, Au, Pb, Bi, Am
Last Acquired: B
Next: Na
-------------- |
|
|
Jackson
Hazard to Others
  
Posts: 189
Registered: 22-5-2018
Location: U S of A
Member Is Offline
Mood:  Happy about new glassware 
|
|
I know. I no longer use it for that reason. I might turn it into boiling chips to get some more use out of it though.
|
|
|
j_sum1
Administrator
       
Posts: 6251
Registered: 4-10-2014
Location: Unmoved
Member Is Offline
Mood: Organised
|
|
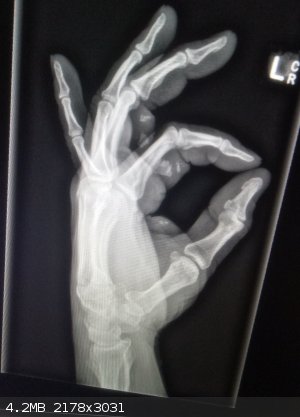
Well I am currently at hospital waiting room with a few bits of beaker in my hand. Not too serious but a bit painful at the moment. Might need
stitches. Certainly needs cleaning.
Full story of my stupidity later...
J.
|
|
|
Ubya
International Hazard
    
Posts: 1247
Registered: 23-11-2017
Location: Rome-Italy
Member Is Offline
Mood: I'm a maddo scientisto!!!
|
|
Quote: Originally posted by j_sum1  | Well I am currently at hospital waiting room with a few bits of beaker in my hand. Not too serious but a bit painful at the moment. Might need
stitches. Certainly needs cleaning.
Full story of my stupidity later...
J. |
hope you can use your hand as soon as possible!
---------------------------------------------------------------------
feel free to correct my grammar, or any mistakes i make
---------------------------------------------------------------------
|
|
|
j_sum1
Administrator
       
Posts: 6251
Registered: 4-10-2014
Location: Unmoved
Member Is Offline
Mood: Organised
|
|
That there is bits of beaker. The orthopedic surgeon is going digging tomorrow.
|
|
|
morganbw
National Hazard
   
Posts: 561
Registered: 23-11-2014
Member Is Offline
Mood: No Mood
|
|
Good luck, looks like it may be sore for a while.
|
|
|
Herr Haber
International Hazard
    
Posts: 1236
Registered: 29-1-2016
Member Is Offline
Mood: No Mood
|
|
A couple of weeks ago I broke a thermometer well that I had forgotten on my bed after unpacking.
This weekend I did it again !
There was only a slight improvement with my procedure as I waited until morning to break the thing that somehow managed to stay with me all night.
Now I have 2 thermometer wells that I can repurpose...

|
|
|
Ubya
International Hazard
    
Posts: 1247
Registered: 23-11-2017
Location: Rome-Italy
Member Is Offline
Mood: I'm a maddo scientisto!!!
|
|
the second one could be used as a gas takeout port
---------------------------------------------------------------------
feel free to correct my grammar, or any mistakes i make
---------------------------------------------------------------------
|
|
|
Herr Haber
International Hazard
    
Posts: 1236
Registered: 29-1-2016
Member Is Offline
Mood: No Mood
|
|
That's a good possibility. In both cases I can seal in place some PTFE or VITON tubing to collect or bubble gas.
|
|
|
j_sum1
Administrator
       
Posts: 6251
Registered: 4-10-2014
Location: Unmoved
Member Is Offline
Mood: Organised
|
|
Quote: Originally posted by j_sum1  | Well I am currently at hospital waiting room with a few bits of beaker in my hand. Not too serious but a bit painful at the moment. Might need
stitches. Certainly needs cleaning.
Full story of my stupidity later...
J. |
Now out of hospital.
I refer you to The Wump Incident.
|
|
|
zed
International Hazard
    
Posts: 2281
Registered: 6-9-2008
Location: Great State of Jefferson, City of Portland
Member Is Offline
Mood: Semi-repentant Sith Lord
|
|
Well, the top front burner on my electric stove, is now shot.
Was I rash, in utilizing it as the heat source, for an impromptu furnace?
I was drilling holes, in what appeared to be 10lb cast iron weight disks. I have lots of extras.
Making ring stands I was.
Drilled my holes in one plate, tapped the holes, and put together a very stable ring stand.
Next plate, just wouldn't drill out. Too hard. At least, I think that is the problem.
Reasoning that the crap iron/steel must have been incidentally hardened by quenching, I decided to anneal the plate. Thereby making it drill-able.
Just put it on the front stove element, covered it with an overturned frying pan, and cranked up the heat.
After a bit, the plate acquired a sort of dull red glow, at which point there was a small explosion and a micro-fire at the base of the heating
element.
I figured the "pop" might be just some cooking oil vapor "dieseling". No big deal. But, now my front stove element has become retarded.
Not heating properly anymore. Oh, well.
https://www.ebay.com/itm/GE-Hotpoint-Kenmore-Range-Cooktop-S...
[Edited on 27-2-2019 by zed]
[Edited on 27-2-2019 by zed]
[Edited on 27-2-2019 by zed]
|
|
|
Herr Haber
International Hazard
    
Posts: 1236
Registered: 29-1-2016
Member Is Offline
Mood: No Mood
|
|
Take the ammonia out of the fridge.
Pour the ammonia in an Erlenmeyer in the sun.
Stopper the Erlenmeyer.
Hear the crashing sound of the stopper falling on the ground.
Oww, another one...
The spirit of adventure was upon me. Having nitric acid and copper, I had only to learn what the words 'act upon' meant. - Ira Remsen
|
|
|
Ubya
International Hazard
    
Posts: 1247
Registered: 23-11-2017
Location: Rome-Italy
Member Is Offline
Mood: I'm a maddo scientisto!!!
|
|
Quote: Originally posted by Herr Haber  | Take the ammonia out of the fridge.
Pour the ammonia in an Erlenmeyer in the sun.
Stopper the Erlenmeyer.
Hear the crashing sound of the stopper falling on the ground.
Oww, another one... |
I hope that only the stopper bit the dust
---------------------------------------------------------------------
feel free to correct my grammar, or any mistakes i make
---------------------------------------------------------------------
|
|
|
TheMrbunGee
Hazard to Others
  
Posts: 364
Registered: 13-7-2016
Location: EU
Member Is Offline
Mood: Phosphorising
|
|
Buchner funnel
Today I confirmed, that round things like nice 70 ml ground glass joint buchner funnel do actually roll on flat surfaces.
On the second part of experiment I learned about gravity, inertia, and properties of different materials, like glass and its literal impact on metal
objects.

Even the ground joint cracked. 
|
|
|
RogueRose
International Hazard
    
Posts: 1590
Registered: 16-6-2014
Member Is Offline
|
|
Are all 600ml beakers suicidal?
I could have sworn this thread was a sticky awhile ago. Was it and was it changed?
I've broken about 6 beakers over the last 4-5 years and 5 of them have been 600ml beakers. What's the deal with these, do they just hate life or
something? Do they feel inferior to the 1000ml and just can no longer go on sitting on the shelf next to it's big brother?
So I was trying to make some liquid soap by adding water to normal potassium based soap and stirring it over low heat on my hot plate. The temp was
set to ~ 2.5, and I don't think it was even hot enough to boil but hot enough to melt the soap and mix it with the water. It got a little thick so I
added ~15-20ml warm water (~110-120F) and some of it touched the side of the beaker after running off the flattish top surface of the soap, and
"CLINK" a nice 3.5" crack up the side and it extended underneath to about of the center of the beaker.
The temp difference couldn't have been more than about 80F, MAYBE 100F, but that even seems high. It was an expensive Pyrex brand beaker I'd used 2x
before and was in perfect shape.
Is it possible that there are counterfeits out there of these things? It seems like an odd thing to counterfeit but I can't imagine real Pyrex
cracking like this at such a low temp diff and so little water coming into contact with the glass. I guess I could see some of these coming from
China with a fake label/brand on them, but they sell boro beakers for a very reasonable price - why not make them generic boro and put Pyrex on them
- or did I maybe just get a beaker with a flaw...
Anyone else seem to have problems with 600ml's?
|
|
|
Sulaiman
International Hazard
    
Posts: 3625
Registered: 8-2-2015
Location: 3rd rock from the sun
Member Is Offline
|
|
Does the beaker have 'PYREX' or 'pyrex' on it ?
'PYREX' is borosilicate, 'pyrex' is toughened soda-lime glass.
'pyrex' is common in USA and also for cafetieres.
see https://en.wikipedia.org/wiki/Pyrex
P.S. I've cracked two borosilicate beakers by using them directly on electric hotplates,
use a sand, salt, water, oil or air bath.
[Edited on 13-10-2019 by Sulaiman]
CAUTION : Hobby Chemist, not Professional or even Amateur
|
|
|
RogueRose
International Hazard
    
Posts: 1590
Registered: 16-6-2014
Member Is Offline
|
|
Quote: Originally posted by Sulaiman  | Does the beaker have 'PYREX' or 'pyrex' on it ?
'PYREX' is borosilicate, 'pyrex' is toughened soda-lime glass.
'pyrex' is common in USA and also for cafetieres.
see https://en.wikipedia.org/wiki/Pyrex
P.S. I've cracked two borosilicate beakers by using them directly on electric hotplates,
use a sand, salt, water, oil or air bath.
[Edited on 13-10-2019 by Sulaiman] |
It's PYREX and I paid a pretty penny for the set of 10 beakers (my most expensive purchase so far) and it was from a science supply company, so it
should be legit unless they are dealing with shady suppliers themselves.
I've dealt with the Pyrex crap many times in my early days, with hot H2SO4 non-the-less (instantaneous shattering at one point). Not fun and sure got
my heart pumping. Good thing it was outside on concrete slab.
As for the hotplate, are you talking about a real ceramic top lab hot plate (with stirrer). Mine's like $650 new (got it for a steal though) so I
would think it should be able to have a beaker on it.. How else am I going to use the stirring? Either way, I was holding the beaker when I poured
the water in and when I moved back to the hotplate the water sloshed A LITTLE, contacted the side and CLINK. The beaker was cool enough I could hold
it with my hand at the top rim so I figured there would be no way that there could be enough temp diff to crack. I haven't even bothered to clean it
out yet I'm so pissed.
[Edited on 10-13-2019 by RogueRose]
|
|
|
Belowzero
Hazard to Others
  
Posts: 173
Registered: 6-5-2020
Location: Member Is Offline
Member Is Offline
|
|
After waiting 24 hours for my filtration to complete (multiple liters of highly concentrated AN solution)
I figured it was time to boil some of the solution down to start the first crystalization round.
My one and only 2L beaker containing 1.5L of the solution was placed on a hotplate and slowly heated.
I waited until it started boiling and after a few small bumbs it formed bubbles and I continued doing other things.
At that point I heard a sizzling sound , at first I thought it was a droplet that hit the hotplate from the side of the glass.
Then some more sizzling, at that point I realized what was going on , my beaker was leaking!!$@!
Hoping to be able to rescue my AN I lifted the beaker from the hotplate and yes you guessed it, the bottom fell out!!
I watched 1.5L of solution hit the hotplate where it instantly formed a densy white smoke while the rest made a mess out of my table and started
leaking on other stuff.
Luckily noone was around to see a large volume of thick dense clouds of smoke leaving the space.
So I wasted nearly a kilo of my precious AN which is annoyingly hard to obtain and destroyed my one and only 2L beaker, what was supposed to be a nice
relaxing session after work turned into a BAD DAY IN THE LAB 
I am glad it wasnt anything dangerous, flamable or worse and eventhough this was a high quality scott duran beaker this can still happen, lessons
learned.
[Edited on 3-7-2020 by Belowzero]
|
|
|
Refinery
Hazard to Others
  
Posts: 371
Registered: 17-2-2014
Member Is Offline
Mood: Still
|
|
I put a fritted filter funnel in an oven in order to drive water out of it.
Few moments later, I hear a series of cracks, like an egg is hatching with glassy sidetone, and right in front of my eyes the funnel cracked right on
the rim of the frit.
It shall serve for now with patent duct tape joint until a new one arrives. I suppose I will dry them with acetone rinse after this.
|
|
|
arkoma
Redneck Overlord
      
Posts: 1761
Registered: 3-2-2014
Location: On a Big Blue Marble hurtling through space
Member Is Offline
Mood: украї́нська
|
|
last week got two one pound bags of strontium nitrate and one pound of hexamine. i was going to combine the two pounds of strontium salt, except i
didn't. yeah, poured a pound of St salt on top of the hexamine. glad i looked before i shook it up. taped it all up tight, and know about where the
dividing line is and will deal with it when i get home (out west) by carefully slicing bag open and cutting into thirds, keeping each end.
*sigh*
"We believe the knowledge and cultural heritage of mankind should be accessible to all people around the world, regardless of their wealth, social
status, nationality, citizenship, etc" z-lib
|
|
|
CharlieA
National Hazard
   
Posts: 646
Registered: 11-8-2015
Location: Missouri, USA
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by Refinery  | I put a fritted filter funnel in an oven in order to drive water out of it.
Few moments later, I hear a series of cracks, like an egg is hatching with glassy sidetone, and right in front of my eyes the funnel cracked right on
the rim of the frit.
It shall serve for now with patent duct tape joint until a new one arrives. I suppose I will dry them with acetone rinse after this.
|
Just out of curiosity, what was the temperature of your oven? Thanks, Charlie
|
|
|
Refinery
Hazard to Others
  
Posts: 371
Registered: 17-2-2014
Member Is Offline
Mood: Still
|
|
I'm not absolutely sure, but I intended to heat it up just to drive out the water, starting from 100C. It cracked within 5 minutes on placing it
there, and it was not on any metal surface or so that it could have been induced by it.
|
|
|
CharlieA
National Hazard
   
Posts: 646
Registered: 11-8-2015
Location: Missouri, USA
Member Is Offline
Mood: No Mood
|
|
I'm just speculating ($5 word for guessing): I would start with the oven at room temperature and raise the heat slowly because I'm guessing that the
frit/glass wall juncture was the problem. Heating at 40* or 50* C(or just a little above room temperature should dry glassware quickly enough. Rinsing
first with acetone is a good idea, but if you then heat it you would want an explosion-proof oven...or maybe just use a blow dryer.
I guess there is also the possibility that the funnel had a small stress crack.
Thanks for the quick reply...us old farts like to talk.
|
|
|
Refinery
Hazard to Others
  
Posts: 371
Registered: 17-2-2014
Member Is Offline
Mood: Still
|
|
If dried with acetone, I would not use oven to dry it because of that. I'm wary of using oven to dry anything that could be flammable. If in a hurry,
I would use heat gun.
I do suspect that it had internal stresses, because it gave up so easily and the breaking line is clean, it looks like it just came apart as two
parts, not that it's broken. No offense, but it was chinese and quite cheap. Very fine looking piece of glassware, in my opinion. Borosilicate glass
should withstand more temperature difference. Anyways, I won't put any fritted filters into oven never again. Too costly to order a new one every now
and then, and too important part of equipment to lose. Filtering pretty much anything under gravity is just painful.
|
|
|
Fyndium
International Hazard
    
Posts: 1192
Registered: 12-7-2020
Location: Not in USA
Member Is Offline
|
|
I broke my best separatory/dropper funnel today while cleaning it. Turned it to rinse and the head hit the edge of the sink and it just snapped off.
Pissed af, but even more for the fact that ordering a new one takes half eternity. There are no local suppliers and the nearest ones takes only few
days less than ordering from china, but with an order of magnitude higher price tag, so it's two bad options for the lesser bad.
|
|
|
| Pages:
1
..
33
34
35
36
37
38 |