VSEPR_VOID
National Hazard
   
Posts: 719
Registered: 1-9-2017
Member Is Offline
Mood: Fullerenes
|
|
Ferrophosphorus General
In the previous thread, (http://www.sciencemadness.org/talk/viewthread.php?tid=83382), it was confirmed by myself and (http://goldrefiningforum.com/~goldrefi/phpBB3/viewtopic.php?...) that the brackets used to hold magnets in conventional hard drives are not
mu-metal, which is mostly nickel, but primarily iron. Branching from this field of ferrous experimentation I am not setting out to try and extract
elemental phosphorus from an alloy containing ~71% iron, ~26% phosphorus, and the rest silicon by mass. I have 1 kilogram of this alloy.
Notes: Phosphorus reacts with nitric acid to form phosphoric acid so it may not be used. Well phosphorus can be prepared from phosphoric acid, it is
very complex and required great temperatures.
I already placed 3.8g of the alloy in a beaker with about 60ml of concentrated hydrochloric acid and could see no reaction at room temperature. The
solution was slightly green but this is most likely due to the regular color of the pool acid.
In once thread a user suggested passing chlorine over ferrophosphorus to yield iron and phosphorus chlorides which could be purified with
distillation. This however has the problem of requiring chlorine gas in large volumes and equipment for the reaction that is resistant to high
temperatures and chlorine.
Within cells interlinked
Within cells interlinked
Within cells interlinked
|
|
|
tetraiodide
Harmless

Posts: 16
Registered: 8-6-2018
Member Is Offline
|
|
Not only that, but several phosphorous chlorides are listed as chemical weapons, and I would personally like to avoid that if it were me. Have you
considered electrolysis? Also one more thing is if it's an alloy, and you remove one component, what remains, I would think, is very finely devided,
and depending on the allotrope that could make pyrophoric/explosive so just somthing is keep in mind. Also I know most people have the gear to do it
but if you could melt the iron distillation would be possible, but at that point you may as well just go get some calcium phosphate and charcoal from
the garden depot. And forget the iron all together.
|
|
|
VSEPR_VOID
National Hazard
   
Posts: 719
Registered: 1-9-2017
Member Is Offline
Mood: Fullerenes
|
|
I like the idea of electrolysis. The resulting iron oxide could be washed away with hydrochloric acid to leave behind fine phosphorus (I would assume
red considering its the most stable allotrope).
I do not have the equipment for high temperature distillation on that scale.
The alloy is in the form of large chunks/crystals which resemble to be meteorites.
Within cells interlinked
Within cells interlinked
Within cells interlinked
|
|
|
wg48
National Hazard
   
Posts: 821
Registered: 21-11-2015
Member Is Offline
Mood: No Mood
|
|
Just in case you don't know: from wiki on iron phosphide: "Iron phosphide reacts with moisture and acids producing phosphine (PH3), a toxic and
pyrophoric gas:
If your ferrophosphorous is brittle you could try crushing it to get a better reaction with acid but that will probably just make more phosphine
faster.
|
|
|
fusso
International Hazard
    
Posts: 1922
Registered: 23-6-2017
Location: 4 ∥ universes ahead of you
Member Is Offline
|
|
Is the ferrophosphorus simply a mixture of Fe & P atoms or the P exist as phosphide ions?
Can P exist as amorphous like carbon?
|
|
|
Fulmen
International Hazard
    
Posts: 1706
Registered: 24-9-2005
Member Is Offline
Mood: Bored
|
|
From what I can find it's in the form of phosphides.
We're not banging rocks together here. We know how to put a man back together.
|
|
|
unionised
International Hazard
    
Posts: 5116
Registered: 1-11-2003
Location: UK
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by tetraiodide  | | Not only that, but several phosphorous chlorides are listed as chemical weapons, and I would personally like to avoid that if it were me. .
|
I don't think they are listed as weapons, but as weapons precursors.
(They are still pretty nasty things to play with)
Iodination of ferrophosphorus might prove interesting.
PI3 would convert alcohols to iodides (as far as I can tell, the presence of FeI2 wouldn't make much difference)
|
|
|
VSEPR_VOID
National Hazard
   
Posts: 719
Registered: 1-9-2017
Member Is Offline
Mood: Fullerenes
|
|
Quote: Originally posted by wg48  | Just in case you don't know: from wiki on iron phosphide: "Iron phosphide reacts with moisture and acids producing phosphine (PH3), a toxic and
pyrophoric gas:
If your ferrophosphorous is brittle you could try crushing it to get a better reaction with acid but that will probably just make more phosphine
faster.
|
I have observed no pyrophoric gasses being evolved when I treated a sample with HCl(aq), HCl(aq) + Peroxide, or when heated.
Further more it does not resemble iron phosphide, but is extremely metallic.
The alloy is resistant to acid. I found it shares this in common with nickel-phosphorus: "High-phosphorus electroless nickel offers high corrosion
resistance, making it ideal for industry standards requiring protection from highly corrosive acidic environments such as oil drilling and coal
mining. With microhardness ranging up to 600 VPN, this type ensures very little surface porosity where pit-free plating is required and is not prone
to staining. Deposits are non-magnetic when phosphorus content is greater than 11.2%"
Source: https://en.wikipedia.org/wiki/Electroless_nickel_plating
[Edited on 9-6-2018 by VSEPR_VOID]
Within cells interlinked
Within cells interlinked
Within cells interlinked
|
|
|
wg48
National Hazard
   
Posts: 821
Registered: 21-11-2015
Member Is Offline
Mood: No Mood
|
|
Quote: Originally posted by VSEPR_VOID  |
I have observed no pyrophoric gasses being evolved when I treated a sample with HCl(aq), HCl(aq) + Peroxide, or when heated.
Further more it does not resemble iron phosphide, but is extremely metallic.
|
I guess your pieces dissolves slowly or very slowly. A powder may be very much faster. Also H2O2 may oxidise phosphine.
Your ferrophosphorous is probably a solid solution or mixture of iron and iron phosphide/s (FeP,Fe2P, Fe3P). Below is a pic of Fe2P. Its looks
metallic.
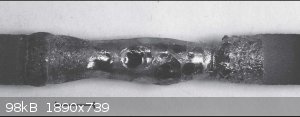
|
|
|
woelen
Super Administrator
        
Posts: 8003
Registered: 20-8-2005
Location: Netherlands
Member Is Offline
Mood: interested
|
|
There are two types of phosphides.
One type has phosphide ions and has well-defined stoichiometry, with phosphorus in oxidation state -3, and reacts easily with acids. Examples are
Zn3P2, Ca3P2, AlP, Mg3P2. With acid these give nearly pure PH3 and they react easily. Some of these even react slowly with water!
The other type is very inert and can have a range of stoichiometries, e.g. Fe2P ... Fe3P, Cu3P, NiP2, and everything inbetween. The oxidation state of
the phosphorus is not well-defined in these cases. These compounds usually are inert and hardly react with acids. I myself have nickel phosphide,
which also looks quite metallic and I did not find anything which reacts with this at room temperature.
Many non-metal elements form such very inert compounds with transition metals, while forming very reactive and well-defined compounds with Ca, Mg, Zn.
Other ones are Si, B, N, C, S.
|
|
|
unionised
International Hazard
    
Posts: 5116
Registered: 1-11-2003
Location: UK
Member Is Offline
Mood: No Mood
|
|
On a related note, does ferrophosphorus react with sulphur?
|
|
|