Succinonitrile
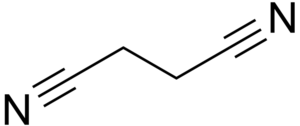 Structure of succinonitrile
| |
| Names | |
|---|---|
| IUPAC name
Butanedinitrile
| |
| Other names
1,2-Dicyanoethane
Ethylene cyanide Ethylene Dicyanide Succinodinitrile | |
| Properties | |
| C4H4N2 | |
| Molar mass | 80.09 g/mol |
| Appearance | Colorless, waxy crystals |
| Odor | Odorless |
| Density | 0.985 g/cm3 (25 °C) |
| Melting point | 54.5 °C (130.1 °F; 327.6 K) |
| Boiling point | 266.1 °C (511.0 °F; 539.2 K) |
| 12.69 g/100 ml (20 °C)[1] | |
| Solubility | Soluble in acetone, chloroform, dioxane, methanol Slightly soluble in benzene, carbon disulfide, diethyl ether, ethanol, hexane |
| Solubility in methanol | 10 g/100 ml |
| Vapor pressure | 0.00778 mmHg at 25 °C |
| Thermochemistry | |
| Std molar
entropy (S |
191.59 J·K−1·mol−1 |
| Std enthalpy of
formation (ΔfH |
139.3–140.4 kJ/mol |
| Hazards | |
| Safety data sheet | Sigma-Aldrich |
| Flash point | 132 °C (270 °F; 405 K) |
| Lethal dose or concentration (LD, LC): | |
| LD50 (Median dose)
|
450 mg/kg (rat, oral) |
| Related compounds | |
| Related compounds
|
Malononitrile Dicyanoacetylene |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Succinonitrile or butanedinitrile, is an organic chemical compound, a nitrile. It has the formula NCCH2CH2CN or C4H4N2.
Contents
Properties
Chemical
Hydrogenation of succinonitrile yields putrescine.
Physical
Succinonitrile is a waxy colorless solid, that slowly turns brownish in air. It is soluble in water and alcohols.
Availability
Butanedinitrile is sold by chemical suppliers.
Preparation
Succinonitrile is mainly produced by the addition of hydrogen cyanide to acrylonitrile:[2]
- CH2=CHCN + HCN → NCCH2CH2CN
Alternatively, it can also be produced by reacting 1,2-dibromoethane or 1,2-diiodoethane with potassium or sodium cyanide.[3][4]
Projects
- Synthesis of putrescine
Handling
Safety
Succinonitrile is expected to be toxic, as it's metabolized to free cyanide which is very toxic. It can be absorbed via ingestion, inhalation and skin/eye contact.[5]
Storage
In closed bottles, away from corrosive reagents.
Disposal
Should be carefully mixed with bleach. Alternatively it can be dissolved in a flammable solvent and burned in an incinerator.
References
- ↑ Yalkowsky, S.H., He, Yan., Handbook of Aqueous Solubility Data: An Extensive Compilation of Aqueous Solubility Data for Organic Compounds Extracted from the AQUASOL dATAbASE. CRC Press LLC, Boca Raton, FL. 2003., p. 82
- ↑ http://www.mrw.interscience.wiley.com/emrw/9783527306732/ueic/article/a17_363/current/html?hd=All%2Csuccinonitrile
- ↑ https://i.imgur.com/6uBH0bE.png
- ↑ https://pubs.acs.org/doi/10.1021/jo01072a600
- ↑ https://www.cdc.gov/niosh/npg/npgd0573.html