Difference between revisions of "Desiccant"
From Sciencemadness Wiki
| (9 intermediate revisions by 3 users not shown) | |||
| Line 7: | Line 7: | ||
*Concentrated [[sulfuric acid]] | *Concentrated [[sulfuric acid]] | ||
*[[Copper(II) sulfate|Copper sulfate]] (anhydrous) | *[[Copper(II) sulfate|Copper sulfate]] (anhydrous) | ||
| − | |||
*[[Magnesium sulfate]] | *[[Magnesium sulfate]] | ||
*[[Phosphorus pentoxide]] | *[[Phosphorus pentoxide]] | ||
| Line 33: | Line 32: | ||
|- | |- | ||
| Activated alumina | | Activated alumina | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Basic or acidic |
| style="text-align: center;"| Medium | | style="text-align: center;"| Medium | ||
| style="text-align: center;"| High | | style="text-align: center;"| High | ||
| Line 54: | Line 53: | ||
|- | |- | ||
| [[Aluminium nitrate]] | | [[Aluminium nitrate]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Slightly acidic |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|No | + | | style="text-align: center;"| No |
| | | | ||
|- | |- | ||
| Bentonite clay | | Bentonite clay | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| style="text-align: center;"| | | style="text-align: center;"| | ||
| style="text-align: center;"| | | style="text-align: center;"| | ||
| Line 82: | Line 67: | ||
|- | |- | ||
| [[Calcium]] | | [[Calcium]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Alkaline |
| style="text-align: center;"| High | | style="text-align: center;"| High | ||
| style="text-align: center;"| Very high | | style="text-align: center;"| Very high | ||
| Line 96: | Line 81: | ||
|- | |- | ||
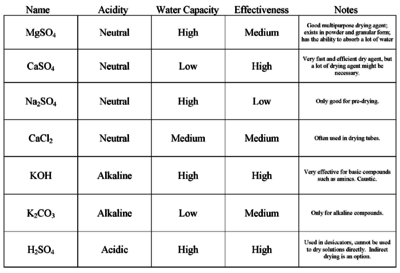
| [[Calcium hydride]] | | [[Calcium hydride]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Alkaline |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|Very high | + | | style="text-align: center;"| Very high |
| − | | style="text-align: center;"|No | + | | style="text-align: center;"| No |
| | | | ||
|- | |- | ||
| [[Calcium nitrate]] | | [[Calcium nitrate]] | ||
| − | | style="text-align: center;"|Neutral | + | | style="text-align: center;"| Neutral |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| | | | ||
|- | |- | ||
| [[Calcium oxide]] | | [[Calcium oxide]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Alkaline |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|No | + | | style="text-align: center;"| No |
| − | | | + | | style="text-align: center;"| Calcium oxide will only remove water from ethanol until 5000 ppm.<ref>Ford, S. G.; Marvel, C. S., Organic Syntheses; Wiley: New York, 1943; Collect. Vol. 11, p 373.</ref> |
|- | |- | ||
| [[Calcium sulfate]] | | [[Calcium sulfate]] | ||
| − | | style="text-align: center;"|Neutral | + | | style="text-align: center;"| Neutral |
| − | | style="text-align: center;"|Low | + | | style="text-align: center;"| Low |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| Very fast and efficient drying agent, but a lot of drying agent might be necessary | | Very fast and efficient drying agent, but a lot of drying agent might be necessary | ||
|- | |- | ||
| − | | Cement (Portland) | + | | [[Portland cement|Cement (Portland)]] |
| − | | style="text-align: center;"|Alkaline | + | | style="text-align: center;"| Alkaline |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|No | + | | style="text-align: center;"| No |
| Used in desiccators, cannot be used directly | | Used in desiccators, cannot be used directly | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
|- | |- | ||
| [[Cobalt(II) chloride]] | | [[Cobalt(II) chloride]] | ||
| Line 155: | Line 119: | ||
| style="text-align: center;"| | | style="text-align: center;"| | ||
| style="text-align: center;"| | | style="text-align: center;"| | ||
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| Mostly used as water indicator | | Mostly used as water indicator | ||
|- | |- | ||
| [[Copper(II) sulfate]] | | [[Copper(II) sulfate]] | ||
| − | | style="text-align: center;"|Neutral | + | | style="text-align: center;"| Neutral |
| − | | style="text-align: center;"|Low | + | | style="text-align: center;"| Low |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| Mostly used as water indicator | | Mostly used as water indicator | ||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
| − | |||
|- | |- | ||
| [[Magnesium]] | | [[Magnesium]] | ||
| style="text-align: center;"| | | style="text-align: center;"| | ||
| style="text-align: center;"| | | style="text-align: center;"| | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|No | + | | style="text-align: center;"| No |
| − | | Reaction is very slow, rarely used | + | | Reaction is very slow, rarely used; Mostly used for removing traces of water |
|- | |- | ||
| [[Magnesium sulfate]] | | [[Magnesium sulfate]] | ||
| − | | style="text-align: center;"|Neutral | + | | style="text-align: center;"| Neutral |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| Good multipurpose drying agent; exists in powder and granular form; has the ability to absorb a lot of water | | Good multipurpose drying agent; exists in powder and granular form; has the ability to absorb a lot of water | ||
|- | |- | ||
| [[Magnesium chloride]] | | [[Magnesium chloride]] | ||
| − | | style="text-align: center;"|Neutral | + | | style="text-align: center;"| Neutral |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| Deliquescent | | Deliquescent | ||
|- | |- | ||
| [[Molecular sieve|Molecular sieves]] | | [[Molecular sieve|Molecular sieves]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Weakly basic |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| − | | | + | | Drying takes hours to days; Unsuitable for drying ketones |
|- | |- | ||
| − | + | | [[Potassium carbonate]] | |
| − | + | | style="text-align: center;"| Acidic | |
| − | + | | style="text-align: center;"| Medium | |
| − | + | | style="text-align: center;"| High | |
| − | + | | style="text-align: center;"| No | |
| − | + | | Good for thoroughly drying predried compounds | |
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | | [[Potassium]] | + | |
| − | | style="text-align: center;"| | + | |
| − | | style="text-align: center;"| | + | |
| − | | style="text-align: center;"| | + | |
| − | | style="text-align: center;"|No | + | |
| − | | | + | |
|- | |- | ||
| [[Potassium carbonate]] | | [[Potassium carbonate]] | ||
| − | | style="text-align: center;"|Alkaline | + | | style="text-align: center;"| Alkaline |
| − | | style="text-align: center;"|Low | + | | style="text-align: center;"| Low |
| − | | style="text-align: center;"|Medium | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| Only for alkaline compounds | | Only for alkaline compounds | ||
|- | |- | ||
| [[Potassium hydroxide]] | | [[Potassium hydroxide]] | ||
| − | | style="text-align: center;"|Alkaline | + | | style="text-align: center;"| Alkaline |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| | | | ||
|- | |- | ||
| [[Silica gel]] | | [[Silica gel]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Weakly acidic |
| − | | style="text-align: center;"| | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Medium |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| | | | ||
|- | |- | ||
| [[Sodium]] | | [[Sodium]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Alkaline |
| − | | style="text-align: center;"| | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Very High |
| − | | style="text-align: center;"|No | + | | style="text-align: center;"| No |
| More often used to remove traces of water from aprotic solvents | | More often used to remove traces of water from aprotic solvents | ||
|- | |- | ||
| [[Sodium hydroxide]] | | [[Sodium hydroxide]] | ||
| − | | style="text-align: center;"|Alkaline | + | | style="text-align: center;"| Alkaline |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| Very effective for basic compounds, such as amines; caustic | | Very effective for basic compounds, such as amines; caustic | ||
|- | |- | ||
| [[Sodium oxide]] | | [[Sodium oxide]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Alkaline |
| − | | style="text-align: center;"| | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Very High |
| − | | style="text-align: center;"|No | + | | style="text-align: center;"| No |
| − | | | + | | More effective when used to dry compounds predried with another desiccant |
|- | |- | ||
| [[Sodium sulfate]] | | [[Sodium sulfate]] | ||
| − | | style="text-align: center;"|Neutral | + | | style="text-align: center;"| Neutral |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|Low | + | | style="text-align: center;"| Low |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| Used to dry solvents; Requires lots of it; only good for predrying; | | Used to dry solvents; Requires lots of it; only good for predrying; | ||
|- | |- | ||
| [[Sulfur trioxide]] | | [[Sulfur trioxide]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Acidic |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|Very high | + | | style="text-align: center;"| Very high |
| − | | style="text-align: center;"|No | + | | style="text-align: center;"| No |
| Tends to form a mist of sulfuric acid in contact with moist air | | Tends to form a mist of sulfuric acid in contact with moist air | ||
|- | |- | ||
| [[Sulfuric acid]] (concentrated) | | [[Sulfuric acid]] (concentrated) | ||
| − | | style="text-align: center;"|Acidic | + | | style="text-align: center;"| Acidic |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"|High | + | | style="text-align: center;"| High |
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Yes, difficult |
| − | | Used in desiccators, cannot be used to dry solutions directly | + | | Used in desiccators, cannot be used to dry solutions directly |
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
| − | + | ||
|- | |- | ||
| [[Zinc chloride]] | | [[Zinc chloride]] | ||
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Acidic |
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Low |
| − | | style="text-align: center;"| | + | | style="text-align: center;"| Low |
| − | | style="text-align: center;"|Yes | + | | style="text-align: center;"| Yes |
| − | | | + | | Regenerating must be done in a stream of hydrogen chloride |
|} | |} | ||
<sup>1</sup>All compounds are considered anhydrous. | <sup>1</sup>All compounds are considered anhydrous. | ||
| Line 509: | Line 242: | ||
[[Category:Desiccants]] | [[Category:Desiccants]] | ||
[[Category:Data tables]] | [[Category:Data tables]] | ||
| + | [[Category:Irritants]] | ||
Latest revision as of 21:08, 3 July 2017
A desiccant is a chemical which is hygroscopic enough to absorb water from hydrated compounds in the same sealed environment.
Common desiccants
- Calcium
- Calcium chloride
- Calcium oxide
- Concentrated sulfuric acid
- Copper sulfate (anhydrous)
- Magnesium sulfate
- Phosphorus pentoxide
- Silica gel
- Sodium and other alkali metals
- Sodium hydroxide
- Sodium oxide
Comparison
| Substance1 | pH | Water capacity | Effectiveness | Reversible | Notes |
|---|---|---|---|---|---|
| Acetonitrile | High | High | Yes | Rarely used | |
| Activated alumina | Basic or acidic | Medium | High | Yes | Can also be used to adsorb fluorides |
| Activated charcoal | Medium | Medium | Yes | Will also adsorb other gasses | |
| Aerogel | High | High | Yes | Expensive | |
| Aluminium nitrate | Slightly acidic | Medium | Medium | No | |
| Bentonite clay | |||||
| Calcium | Alkaline | High | Very high | No | Reaction with water releases large amounts of hydrogen |
| Calcium chloride | Neutral | High | Medium | Yes | Deliquescent; often used in drying tubes |
| Calcium hydride | Alkaline | High | Very high | No | |
| Calcium nitrate | Neutral | Medium | Medium | Yes | |
| Calcium oxide | Alkaline | High | High | No | Calcium oxide will only remove water from ethanol until 5000 ppm.[1] |
| Calcium sulfate | Neutral | Low | High | Yes | Very fast and efficient drying agent, but a lot of drying agent might be necessary |
| Cement (Portland) | Alkaline | Medium | Medium | No | Used in desiccators, cannot be used directly |
| Cobalt(II) chloride | Yes | Mostly used as water indicator | |||
| Copper(II) sulfate | Neutral | Low | Medium | Yes | Mostly used as water indicator |
| Magnesium | High | No | Reaction is very slow, rarely used; Mostly used for removing traces of water | ||
| Magnesium sulfate | Neutral | High | Medium | Yes | Good multipurpose drying agent; exists in powder and granular form; has the ability to absorb a lot of water |
| Magnesium chloride | Neutral | High | Medium | Yes | Deliquescent |
| Molecular sieves | Weakly basic | High | High | Yes | Drying takes hours to days; Unsuitable for drying ketones |
| Potassium carbonate | Acidic | Medium | High | No | Good for thoroughly drying predried compounds |
| Potassium carbonate | Alkaline | Low | Medium | Yes | Only for alkaline compounds |
| Potassium hydroxide | Alkaline | High | High | Yes | |
| Silica gel | Weakly acidic | High | Medium | Yes | |
| Sodium | Alkaline | High | Very High | No | More often used to remove traces of water from aprotic solvents |
| Sodium hydroxide | Alkaline | High | High | Yes | Very effective for basic compounds, such as amines; caustic |
| Sodium oxide | Alkaline | High | Very High | No | More effective when used to dry compounds predried with another desiccant |
| Sodium sulfate | Neutral | High | Low | Yes | Used to dry solvents; Requires lots of it; only good for predrying; |
| Sulfur trioxide | Acidic | High | Very high | No | Tends to form a mist of sulfuric acid in contact with moist air |
| Sulfuric acid (concentrated) | Acidic | High | High | Yes, difficult | Used in desiccators, cannot be used to dry solutions directly |
| Zinc chloride | Acidic | Low | Low | Yes | Regenerating must be done in a stream of hydrogen chloride |
1All compounds are considered anhydrous.
Gallery
- ↑ Ford, S. G.; Marvel, C. S., Organic Syntheses; Wiley: New York, 1943; Collect. Vol. 11, p 373.