Alcohol
 |
This article is a stub. Please help Sciencemadness Wiki by expanding it, adding pictures, and improving existing text.
|
Alcohols are organic compounds which contain a hydroxyl functional group (-OH) bonded to a saturated carbon atom.
Contents
Types of Alcohols
Alcohols are divided according to the type of carbon atom to which the hydroxyl functional group is bonded:
- Primary alcohols
- Primary alcohols are ones in which the carbon atom that is bonded to a hydroxyl functional group is bounded to only one other carbon atom.
- Primary alcohols are first oxidized to aldehydes, then to carboxylic acids.
- Secondary alcohols
- Secondary alcohols are ones in which the carbon atom that is bonded to a hydroxyl functional group is bounded to two other carbon atom.
- Secondary alcohols are oxidized to ketones.
- Tertiary alcohols
- Tertiary alcohols are ones in which the carbon atom that is bonded to a hydroxyl functional group is bounded to three other carbon atom.
- Tertiary alcohols cannot be oxidized.
Nomenclature
Systematic names
Common names
| Chemical Formula | IUPAC Name | Common Name |
|---|---|---|
| Monohydric alcohols | ||
| CH3OH | Methanol | Wood alcohol |
| C2H5OH | Ethanol | Alcohol |
| C3H7OH | 2-propanol | Rubbing alcohol |
| C4H9OH | 1-Butanol | Butanol |
| C5H11OH | 1-Pentanol | Amyl alcohol |
| C16H33OH | Hexadecan-1-ol | Cetyl alcohol |
| Polyhydric alcohols | ||
| C2H4(OH)2 | Ethane-1,2-diol | Ethylene glycol |
| C3H6(OH)2 | Propane-1,2-diol | Propylene glycol |
| C3H5(OH)3 | Propane-1,2,3-triol | Glycerol |
| C4H6(OH)4 | Butane-1,2,3,4-tetraol | Erythritol, Threitol |
| C5H7(OH)5 | Pentane-1,2,3,4,5-pentol | Xylitol |
| C6H8(OH)6 | Hexane-1,2,3,4,5,6-hexol | Mannitol, Sorbitol |
| C7H9(OH)7 | Heptane-1,2,3,4,5,6,7-heptol | Volemitol |
| Unsaturated aliphatic alcohols | ||
| C3H5OH | Prop-2-ene-1-ol | Allyl alcohol |
| C10H17OH | 3,7-Dimethylocta-2,6-dien-1-ol | Geraniol |
| C3H3OH | Prop-2-in-1-ol | Propargyl alcohol |
| Alicyclic alcohols | ||
| C6H6(OH)6 | Cyclohexane-1,2,3,4,5,6-hexol | Inositol |
| C10H19OH | 2 - (2-propyl)-5-methyl-cyclohexane-1-ol | Menthol |
Physical and chemical properties
Applications
Preparation
Methanol can be obtained from the destructive distillation of wood, though formaldehyde is also produced in this process.
Ethanol can be obtained from the fermentation of sugar.
Reactions
Oxidation
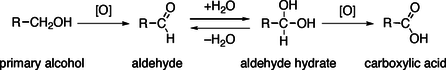
Primary alcohols can be oxidized either to aldehydes or to carboxylic acids, while the oxidation of secondary alcohols normally terminates at the ketone stage. Tertiary alcohols are resistant to oxidation.
The direct oxidation of primary alcohols to carboxylic acids normally proceeds via the corresponding aldehyde, which is transformed via an aldehyde hydrate by reaction with water before it can be further oxidized to the carboxylic acid.
Reagents useful for the transformation of primary alcohols to aldehydes are normally also suitable for the oxidation of secondary alcohols to ketones. These include Collins reagent and Dess-Martin periodinane. The direct oxidation of primary alcohols to carboxylic acids can be carried out using potassium permanganate or the Jones reagent.
Esterification
The esterification is a reaction of an alcohol with an acid, the result is an ester.
Dehydration
Dehydration is a process of separating all the water molecules from the alcohol. This is done with a dehydrating agent which is more hygroscopic than the alcohol, one such example are the alkali metals, dehydrated calcium chloride or anhydrous calcium oxide (quicklime)
This can also be done with a 3A molecular sieves.
Deprotonation
Deprotonation of alcohols by superbases or active metals gives salt-like compounds called alkoxides. Because most alcohols are weaker acids than water, alkoxides are typically hydrolyzed fully and irreversibly into the original alcohol and a hydroxide. Only certain alicyclic keto-alcohols such as rhodizonic acid are acidic enough to make the corresponding alkoxides stable in aqueous solution.
Nucleophilic substitution
Safety
Toxicity
Methanol is poisonous, and ingestion may lead to blindness and death. Longer chain alcohols, such as ethanol, n-propanol and isopropanol, are much less toxic. Higher alcohols, such as butanol, pentanol, and their isomers also display low toxicity.
Except for ethylene glycol, most polyols have low toxicity.
Monohydric alcohols can be absorbed through skin.