Aminoguanidinium bicarbonate
 Aminoguanidine bicarbonate prepared from calcium cyanamide
| |
| Names | |
|---|---|
| IUPAC name
2-Aminoguanidinium hydrogen carbonate
| |
| Other names
Aminoguanidine hydrocarbonate
Aminoguanidine hydrogencarbonate Aminoguanidinium hydrogen carbonate Guanylhydrazine hydrogen carbonate | |
| Properties | |
| C2H8N4O3 | |
| Molar mass | 136.11 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.56 g/cm3 (20 °C) |
| Melting point | 170–172 °C (338–342 °F; 443–445 K) |
| Boiling point | Decomposes |
| 0.27 g/100 ml (20 °C) 0.33 g/100 ml (30 °C) | |
| Vapor pressure | ~0 mmHg |
| Hazards | |
| Safety data sheet | Sigma-Aldrich |
| Lethal dose or concentration (LD, LC): | |
| LD50 (Median dose)
|
5.000 mg/kg (rat, oral) 1.160 mg/kg (rat, IP) |
| Related compounds | |
| Related compounds
|
Aminoguanidinium nitrate |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Aminoguanidinium bicarbonate or aminoguanidine bicarbonate is a chemical compound used as precursor for the preparation of aminoguanidine compounds. It has the chemical formula C2H8N4O3.
Contents
Properties
Chemical
Addition of an equimolar amount of freebase aminoguanidine to aminoguanidine bicarbonate will yield aminoguanidinium carbonate.
Aminoguanidinium bicarbonate will react with acids to yield their respective salts.
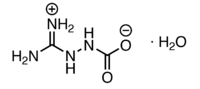
X-ray analysis has shown that solid aminoguanidine bicarbonate is actually a zwitterionic molecule, 2-guanidinium-1-aminocarboxylate monohydrate.[1]
Physical
Aminoguanidinium bicarbonate is a white solid, slightly soluble in water. Recrystallization from hot water is possible, but some decomposition always occurs and reprecipitation tends to be slow and incomplete.
Availability
Aminoguanidine bicarbonate is sold by chemical suppliers.
Preparation
Aminoguanidinium bicarbonate can be prepared by reacting calcium cyanamide with hydrazine sulfate.
It can also be easily prepared by reducing nitroguanidine with zinc powder. Diachrynic used this route obtaining a great yield:[2]
41.14 g zinc powder (629 mmol, 3.3 molar eq.) are weighed and put to the side. Into a reaction flask of at least 500 mL are put 20.00 g nitroguanidine (192 mmol, 1 molar eq.) and 47.62 g of ammonium sulfate (360 mmol, 1.88 molar eq.) in 285 mL of water. The suspension is stirred and not everything dissolves, this is expected. The reaction flask is immersed in an ice bath and equipped with magnetic stirring and a thermometer. Stirring is started. Once the solution reaches 10 °C the addition of small spatulas of zinc powder at a time is started. Monitor the exotherm and don't add too much at once, however the reaction is fairly easy to control. 3-4 spatulas of zinc can be added at a time, making the temperature jump up by 5-8 °C. The reaction was kept between 5-15 °C, leaning to the latter temperature. The complete addition of zinc took about 1 hour, during which the ice bath was refilled just once. Afterwards the reaction was left stirring at about 15 °C for a further 30 minutes. The pH rose to about 8-9. Using a fritted vacuum filter the zinc oxide sludge was removed, it filtered fairly easily. The yellow colored filtrate is put into a flask with magnetic stirring, and 8.57 g of 25% ammonia solution (126 mmol, 0.66 molar eq.) are added as well as 28.57 g of sodium bicarbonate (340 mmol, 0.94 molar eq.) with stirring, it dissolves after a short while. The solution is left standing for 12 h during which the aminoguanidine bicarbonate slowly precipitates. Afterwards the product is vacuum filtered off and air dried.
Yield of aminoguanidine bicarbonate: 15.700 g (115 mmol, 60% based on nitroguanidine)
Projects
- Preparation of aminoguanidine compounds and complexes
- Preparation of tetrazoles
Handling
Safety
Aminoguanidine compounds don't pose significant toxicity, though they should still be handled with proper protection.
Storage
In closed bottles.
Disposal
Dilute it with water and pour it down the drain.
Gallery
References
- ↑ Kolev, T., & Petrova, R. 2003. Zwitterionic 2-guanidinium-1-aminocarboxylate monohydrate. Acta Crystallographica Section E Structure Reports Online, 59(4), 447–449. doi:10.1107/s1600536803004203
- ↑ https://www.sciencemadness.org/whisper/viewthread.php?tid=155675