Methyl cellosolve
From Sciencemadness Wiki
 |
This article is a stub. Please help Sciencemadness Wiki by expanding it, adding pictures, and improving existing text.
|
| |
| Names | |
|---|---|
| IUPAC name
2-Methoxyethanol
| |
| Systematic IUPAC name
2-Methoxyethanol | |
| Other names
Ethylene glycol monomethyl ether
EGME Methyl cellosolve | |
| Identifiers | |
| 109-86-4 | |
| Jmol-3D images | Image |
| |
| Properties | |
| C3H8O2 | |
| Molar mass | 76.09 g/mol |
| Appearance | Colorless liquid |
| Odor | Ethereal |
| Density | 0.965 g/cm3 |
| Melting point | −85 °C (−121 °F; 188 K) |
| Boiling point | 124 to 125 °C (255 to 257 °F; 397 to 398 K) |
| Miscible | |
| Solubility | Miscible with alcohols |
| Vapor pressure | 6 mmHg (20 °C) |
| Hazards | |
| Safety data sheet | Sigma-Aldrich |
| Flash point | 39 °C (102 °F; 312 K) |
| Related compounds | |
| Related compounds
|
Ethylene glycol |
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
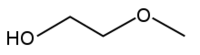
2-Methoxyethanol, also known as ethylene glycol monomethyl ether or methyl cellosolve, is a common solvent.
Contents
Properties
Chemical
Methyl cellosolve is both a primary alcohol and an ether.
Physical
Methyl cellosolve is a colorless liquid, with an ethereal odor. It is miscible with water and most organic solvents.
Availability
Many OTC paint and dye solvents have this compound in their composition. Fractional distillation is required to isolate the compound.
Preparation
Industrially, methyl cellosolve is produced by the reaction of methanol with ethylene oxide.
Projects
- Paint stripper
Handling
Safety
Methyl cellosolve is an endocrine disruptor. It is toxic to the bone marrow and testes.
Storage
2-Methoxyethanol should be kept in closed bottles. BHT is sometimes added as stabilizer.[1]
Disposal
Incineration outside should suffice.
References
Relevant Sciencemadness threads
Categories:
- Article stubs
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Chemical compounds
- Organic compounds
- Alcohols
- Ethers
- Liquids